2112. Chloroacetic Acid
Nomenclature
CAS number: 79-11-8
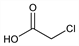
2-Chloroacetic acid; chloroethanoic acid; monochloroacetic acid; MCA. C2H3ClO2; mol wt 94.50.
C 25.42%, H 3.20%, Cl 37.52%, O 33.86%.
Description and references
Made by chlorination of glacial acetic acid in presence of small amount of sulfur or iodine; also by hydrolysis of trichlorethylene with 90% H2SO4. Lab prepn: Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin, 40th ed., 1961) pp 109, 110. Manuf: Faith, Keyes & Clark's Industrial Chemicals, F. A. Lowenheim, M. K. Moran, Eds. (Wiley-Interscience, New York, 4th ed., 1975) pp 254-257. Toxicity: Woodward et al., J. Ind. Hyg. Toxicol. 23, 78 (1941).
Properties
Colorless or white, deliquesc crystals. d 1.580. Exists in α, β and γ forms having mp 63°, 55-56° and 50° respectively. mp for acid of commerce is 61-63°. bp for all forms is 189°. Very sol in water; sol in alcohol, benzene, chloroform, ether. Poisonous, corrosive. Keep well closed and in a cool place.Derivative
Sodium salt.Nomenclature
CAS number: 3926-62-3
Somon (Atlas). C2H2ClNaO2; mol wt 116.48.
C 20.62%, H 1.73%, Cl 30.44%, Na 19.74%, O 27.47%.