2167. Chlorothen
Nomenclature
CAS number: 148-65-2
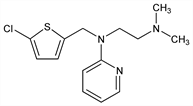
N-[(5-Chloro-2-thienyl)methyl]-N′,N′-dimethyl-N-2-pyridinyl-1,2-ethanediamine; 2-[(5-chloro-2-thenyl)(2-dimethylaminoethyl)amino]pyridine; N,N-dimethyl-N′-(2-pyridyl)-N′-(5-chloro-2-thenyl)ethylenediamine; N,N-dimethyl-N′-(α-pyridyl)-N′-(2-methyl-5-chlorothienyl)ethylenediamine; N-5-chloro-2-thienylmethyl-N′,N′-dimethyl-N-2-pyridylethylenediamine; chloropyrilene; chloromethapyrilene; chlorothenylpyramine. C14H18ClN3S; mol wt 295.83.
C 56.84%, H 6.13%, Cl 11.98%, N 14.20%, S 10.84%.
Description and references
Prepd by the condensation of 5-chloro-2-thenyl chloride and N,N-dimethyl-N′-(2-pyridyl)ethylenediamine in the presence of sodium or potassium amide: R. C. Clapp et al., J. Am. Chem. Soc. 69, 1549 (1947); L. P. Kyrides, US 2581869 (1952 to Monsanto). Toxicity data: J. C. Castillo et al., J. Pharmacol. Exp. Ther. 96, 388 (1949).
Properties
Liquid. bp1.0 155-156°. Strong base.Derivative
Hydrochloride.Nomenclature
Thenclor.C14H18ClN3S.HCl; mol wt 332.29.
C 50.60%, H 5.76%, Cl 21.34%, N 12.65%, S 9.65%.
Properties
Crystals, mp 106-108°. Freely sol in water.Derivative
Citrate.Nomenclature
CAS number: 148-64-1
Tagathen (Lederle). C14H18ClN3S.C6H8O7; mol wt 487.95.
C 49.23%, H 5.37%, Cl 7.27%, N 8.61%, S 6.57%, O 22.95%.
Properties
Crystals, mp 112-116°. On further heating gradually solidifies and remelts 125-140° (dec). uv max: 240 nm (E1%1cm 390-410). One gram dissolves in 35 ml water, in about 65 ml alc. Practically insol in ether, chloroform, benzene. pH of 1% aq soln 3.9 to 4.1. LD50 i.p. in mice: 105 mg/kg (Castillo).Therapeutic Category
Antihistaminic.
Keywords
Antihistaminic; Ethylenediamine Derivatives