2166. Chlorothalonil
Nomenclature
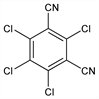
CAS number: 1897-45-6
2,4,5,6-Tetrachloro-1,3-benzenedicarbonitrile; tetrachloroisophthalonitrile; m-tetrachlorophthalodinitrile; 2,4,5,6-tetrachloro-1,3-dicyanobenzene; 1,3-dicyano-2,4,5,6-tetrachlorobenzene; chlorthalonil; DAC-2787; Daconil 2787 (Syngenta); Bravo (Syngenta). C8Cl4N2; mol wt 265.91.
C 36.13%, Cl 53.33%, N 10.53%.
Description and references
Fungicidal properties first described by N. J. Turner et al., Contrib. Boyce Thompson Inst. 22, 303 (1964). Prepn: R. D. Battershell, H. Bluestone, US 3290353 (1966 to Diamond Alkali); R. M. Bimber, US 3652637 (1972 to Diamond Shamrock). Toxicity studies in mice: H. Yoshikawa, K. Kawai, Ind. Health 4, 11 (1966). Review of carcinogenic risk: IARC Monographs 30, 319-328 (1983).