2186. Chlorpropamide
Nomenclature
CAS number: 94-20-2
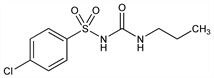
4-Chloro-N-[(propylamino)carbonyl]benzenesulfonamide; 1-(p-chlorophenylsulfonyl)-3-propylurea; 1-(p-chlorobenzenesulfonyl)-3-propylurea; N-propyl-N′-(p-chlorobenzenesulfonyl)urea; P-607; Diabinese (Pfizer); Insogen (Altana). C10H13ClN2O3S; mol wt 276.74.
C 43.40%, H 4.73%, Cl 12.81%, N 10.12%, O 17.34%, S 11.59%.
Description and references
Oral sulfonylurea hypoglycemic agent. Prepn: F. J. Marshall, M. V. Sigal, Jr., J. Org. Chem. 23, 927 (1958); GB 853555; W. M. McLamore, US 3349124 (1960, 1967 both to Pfizer); V. J. Bauer et al., J. Org. Chem. 31, 3440 (1960). Pharmacology and metabolism: Khurana et al., Indian J. Med. Res. 55, 1084 (1967); Brotherton et al., Clin. Pharmacol. Ther. 10, 505 (1969); Madsen et al., Eur. J. Pharmacol. 13, 374 (1971). Toxicity study: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). HPLC and GC-MS determn in horse plasma as a screen for horse racing: H. C. Chau et al, J. Chromatogr. B 712, 243 (1998).
Properties
Crystals from dil ethanol, mp 127-129°. uv max (0.01N HCl): 232.5 nm (ε 16500). Soly in water at pH 6: 2.2 mg/ml. Practically insol at pH 7.3. Sol in alc; moderately sol in chloroform; sparingly sol in ether, benzene. LD50 i.p. in rats: 580 mg/kg (Goldenthal).Therapeutic Category
Antidiabetic.
Keywords
Antidiabetic; Sulfonylurea Derivatives