2185. Chlorpromazine
Nomenclature
CAS number: 50-53-3
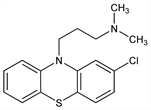
2-Chloro-N,N-dimethyl-10H-phenothiazine-10-propanamine; 2-chloro-10-(3-dimethylaminopropyl)phenothiazine; 3-chloro-10-(3-dimethylaminopropyl)phenothiazine; N-(3-dimethylaminopropyl)-3-chlorophenothiazine; 2601-A; HL-5746; RP-4560; SKF-2601-A. C17H19ClN2S; mol wt 318.86.
C 64.03%, H 6.01%, Cl 11.12%, N 8.79%, S 10.06%.
Description and references
Prepn: Charpentier et al., Compt. Rend. 235, 59 (1952); Charpentier, US 2645640 (1953 to Rh^one-Poulenc). Effects of neuroleptics on dopamine receptors: N.-E. Anden et al., Eur. J. Pharmacol. 11, 303 (1970). Toxicity study: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Review of analytical methods for determn in pharmaceutical prepns: L. F. S. Chagonda, J. S. Millership, J. Pharm. Biomed. Anal. 7, 271-278 (1989). Brief historical review: G. Curzon, Trends Pharmacol. Sci. 11, 61-63 (1990).
Properties
Oily liq. Amine odor. Alkaline reaction. bp0.8 200-205°.Derivative
Hydrochloride.Nomenclature
CAS number: 69-09-0
Ampliactil (Aventis); Amplictil (Aventis); Chloractil (DDSA); Chlorazin (Streuli); Contomin (Takeda); Fenactil (Jelfa); Hibernal (Aventis); Largactil (Aventis); Plegomazin (Servier); Promacid (CID); Promactil (Combiphar); Propaphenin (Rodleben); Prozin (Lusofarmaco); Taroctyl (Taro); Thorazine (GSK); Wintermin (Shionogi). C17H19ClN2S.HCl; mol wt 355.33.
C 57.46%, H 5.67%, Cl 19.95%, N 7.88%, S 9.02%.
Properties
Crystals, dec 179-180° (capillary); 194-196° (microblock). uv curve: Neuhoff, Auterhoff, Arch. Pharm. 288, 400 (1955). pH of 5% aq soln 4.0-5.5. One gram dissolves in 2.5 ml water. Sol in methanol, ethanol, chloroform. Practically insol in ether, benzene. Slightly acid to litmus. LD50 orally in rats: 225 mg/kg (Goldenthal).Therapeutic Category
Antiemetic; antipsychotic.
Therapeutic Category (Veterinary)
Antiemetic; tranquilizer.Keywords
Antiemetic; Antipsychotic; Phenothiazines