2192. Chlortetracycline
Nomenclature
Description and references
Antibiotic substance isolated from the substrate of Streptomyces aureofaciens: Duggan, Ann. N.Y. Acad. Sci. 51, 177 (1948); US 2482055 (1949 to Am. Cyanamid); Broschard et al., Science 109, 199 (1949). Structure: Stephens et al., J. Am. Chem. Soc. 74, 4976 (1952); 76, 3568 (1954). Crystal structure: Donohue et al., ibid. 85, 851 (1963). Absolute configuration: Dobrynin et al., Tetrahedron Lett. 1962, 901. Purification: Winterbottom et al., US 2899422 (1959 to Am. Cyanamid). Improved process: Miller et al.; Goodman, US 2987449; US 3050446 (1961, 1962 to Am. Cyanamid). Toxicity: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Comprehensive description: G. Schwartzman et al., Anal. Profiles Drug Subs. 8, 101-137 (1979).
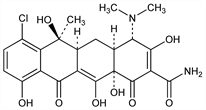
Properties
Golden-yellow crystals, mp 168-169°. [α]D23 -275.0° (methanol). uv max (0.1N HCl): 230, 262.5, 367.5 nm; (0.1N NaOH): 255, 285, 345 nm. Soly in water: 0.5-0.6 mg/ml. Very sol in aq solns above pH 8.5. Freely sol in the Cellosolves, dioxane, and Carbitol. Slightly sol in methanol, ethanol, butanol, acetone, ethyl acetate, benzene. Practically insol in ether, petr ether.Derivative
Crystal salt.Properties
White powder. Insol in water. Marketed as suspension preserved with methyl and propyl p-hydroxybenzoate; stable and fully active for at least one year at room temps.Derivative
Hydrochloride.Nomenclature
Properties
Bitter, yellow rhomboid crystals. Dec above 210°. [α]D23 -240°. Soly at about 28° (mg/ml): water 8.6; methanol 17.4; ethanol 1.7: Weiss et al., Antibiot. Chemother. 7, 374 (1957). Sol in solns of alkali hydroxides and carbonates. Practically insol in acetone, ether, chloroform, dioxane. pH of satd aq soln 2.8-2.9. LD50 orally in rats: 10300 mg/kg (Goldenthal).Therapeutic Category
Antibacterial; antiamebic.
Therapeutic Category (Veterinary)
Antimicrobial.Keywords
Antiamebic; Antibacterial (Antibiotics); Tetracyclines