Nomenclature
CAS number: 57-88-5
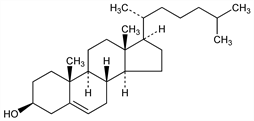
(3β)-Cholest-5-en-3-ol; cholesterin.
C
27H
46O; mol wt 386.65.
C 83.87%, H 11.99%, O 4.14%.
Description and references
Principal sterol of the higher animals. Found
in all body tissues, esp in the brain, spinal cord, and in animal
fats or oils. Main constituent of gallstones. Prepd commercially
from the spinal cord of cattle by petr ether extraction of the nonsaponifiable
matter. Also produced from wool grease. Cholesterol from animal
organs always contains cholestanol (dihydrocholesterol) and other
satd sterols. Purification by repeated bromination: Schoenheimer, J. Biol. Chem. 105, 355 (1934);
Fieser, Org. Synth. coll.
vol. IV, 195 (1963). Laboratory procedure for isoln from gallstones:
L. F. Fieser, Organic Experiments (Heath, Boston, 3rd ed., 1964) p 70. Total synthesis: Keana, Johnson, Steroids 4, 457 (1964). Reviews
and bibliographies: Fieser, Fieser, Steroids (Reinhold, New York, Chapman & Hall, London, 1959); Lettré et al., Ueber Sterine, Gallens"auren
und verwandte Naturstoffe (Stuttgart, 2nd ed., 1955);
R. P. Cook, Cholesterol (Chemistry, Biochemistry and Pathology) (Academic Press, New York, 1958) 542 pp; J. T. Gwynne, J. F. Strauss, Endocr. Rev. 3, 299-329 (1982).
Properties
Monohydrate, pearly leaflets or plates from dil
alcohol. Becomes anhydr at 70-80°. When anhydr mp 148.5°. Has been sublimed as
orthorhombic needles. bp0.5 233°; bp760 360° (some decompn). d 1.03 (monohydrate); d1919 1.052 (anhydr). [α]D20 -31.5° (c = 2 in ether); [α]D20 -39.5° (c = 2 in
chloroform). Absorption spectrum: Heilbron et al., J. Chem. Soc. 1928, 47. Practically insol in water (about 0.2 mg/100 ml H2O). Slightly sol in alc (1.29% w/w at 20°), more sol in hot alc
(100 g of satd 96% alcoholic soln contains 28 g at 80°). One gram
dissolves in 2.8 ml ether, in 4.5 ml chloroform, in 1.5 ml pyridine.
Also sol in benzene, petr ether, oils, fats. Soly in aq solns of
bile salts: Rosin, Z. Physiol. Chem. 124, 282 (1923). Solubilization: Gemant, Life Sci. 1, 233 (June 1962).
Is pptd by digitonin. Gives intense red color with rosaniline in
chloroform soln.Derivative
Methyl ether.
C
28H
48O; mol wt 400.68.
C 83.93%, H 12.07%, O 3.99%.
Properties
Crystals from acetone, mp 84°. [α]D20 -45.8° (c = 1.2 in chloroform).Derivative
Acetate.
C
29H
48O
2; mol wt 428.69.
C 81.25%, H 11.29%, O 7.46%.
Properties
Needles from acetone, mp 115-116°. [α]D20 -47.4° (c = 2 in chloroform).Derivative
Benzoate.
C
34H
50O
2; mol wt 490.76.
C 83.21%, H 10.27%, O 6.52%.
Properties
mp 145.5° (the melt becomes clear at 180°). [α]D25 -13.7° (c = 0.9
in chloroform).Use
Pharmaceutic aid (emulsifying agent).
