2272. Cilastatin
Nomenclature
Description and references
Reversible dehydropeptidase I inhibitor; prevents renal metabolism of penem and carbapenem antibiotics. Prepn: D. W. Graham et al., EP 48301(1982 to Merck & Co.); and activity: idem et al., J. Med. Chem. 30, 1074 (1987). Combination with thienamycins: H. Kropp et al., EP 48025; F. M. Kahan, H. Kropp, US 4539208 (1982, 1985 both to Merck & Co.). HPLC determn in serum: C. M. Myers, J. L. Blumer, Antimicrob. Agents Chemother 26, 78 (1984). Effect on imipenem pharmacokinetics: S. R. Norrby et al., ibid. 23, 300 (1983); on intrathecal penetration of imipenem: A. W. Chow et al., ibid. 23, 634 (1983). Series of articles on pharmacokinetics, tolerance, and efficacy of combination with imipenem: J. Antimicrob. Chemother. 12, Suppl. D, 1-155 (1983); Infection 14, Suppl. 2, S111-S180 (1986). Review of clinical experience in serious infections: J. A. Balfour et al., Drugs 51, 99-136 (1996); of nephroprotective effect with cyclosporin: A. Tejedor et al., Curr. Med. Res. Opin. 23, 505-513 (2007).
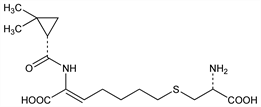
Properties
Amorphous solid. [α]D25 +17.6° (c = 0.5 in methanol). [α]D25 +14.2° (c = 0.5 in 0.1N HCl).Derivative
Sodium salt.Nomenclature
Properties
Off-white to yellowish-white hygroscopic, amorphous solid. pKa1 2.0; pKa2 4.4; pKa3 9.2. Very sol in water, methanol.Derivative
Mixture of sodium salt with imipenem see Imipenem.Therapeutic Category
Antibacterial adjunct (dipeptidase inhibitor).
Keywords
Antibacterial Adjuncts; Renal Dipeptidase Inhibitors