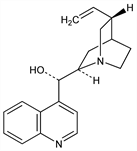
2287. Cinchonine
Nomenclature
CAS number: 118-10-5
(9S)-Cinchonan-9-ol. C19H22N2O; mol wt 294.39.
C 77.52%, H 7.53%, N 9.52%, O 5.43%.
Description and references
Occurs in most varieties of cinchona bark, especially in bark of Cinchona micrantha R. & P., Rubiaceae. Isoln and structure: Rabe, Ber. 41, 63 (1908). Stereoisomeric with cinchonidine: Koenigs, Ann. 347, 182 (1906). Configuration: Prelog, Zalán, Helv. Chim. Acta 27, 535 (1944); Prelog, H"afliger, ibid. 33, 2021 (1950); Roth, Pharmazie 16, 257 (1961); Lyle, Keefer, Tetrahedron 23, 3253 (1967). Crystal and molecular structure: B. Oleksyn et al., Acta Crystallogr. B35, 440 (1979). Biosynthetic studies: Battersby, Parry, Chem. Commun. 1971, 31. Toxicity study: C. C. Johnson, C. F. Poe, Acta Pharmacol. Toxicol. 4, 265 (1949).
Properties
Prisms, needles from alcohol or ether. mp about 265°; begins to sublime at 220°. [α]D +229° (alcohol). uv absorption data: Kamath et al., Indian J. Chem. 6, 510 (1968). One gram dissolves in 60 ml alcohol, 25 ml boiling alcohol, 110 ml chloroform, 500 ml ether. Practically insol in water. pK1 5.85, pK2 9.92. Protect from light. LD50 i.p. in rats: 152 mg/kg (Johnson, Poe).Derivative
Dihydrochloride.C19H22N2O.2HCl; mol wt 367.31.
C 62.13%, H 6.59%, N 7.63%, O 4.36%, Cl 19.30%.
Properties
White or faintly yellow crystals or crystalline powder. Freely sol in water or alcohol. Protect from light.Derivative
Hydrochloride dihydrate.C19H22N2O.HCl.2H2O; mol wt 366.88.
C 62.20%, H 7.42%, N 7.64%, O 13.08%, Cl 9.66%.
Properties
Fine crystals. mp when anhydr about 215° with dec. One gram dissolves in 20 ml water, 3.5 ml boiling water, 1.5 ml alcohol, 20 ml chloroform; slightly sol in ether. The aq soln is practically neutral. Protect from light.Derivative
Sulfate dihydrate.(C19H22N2O)2.H2SO4.2H2O; mol wt 722.89.
C 63.14%, H 6.97%, N 7.75%, O 17.71%, S 4.44%.
Properties
Lustrous, very bitter crystals. mp when anhydr about 198°. One gram dissolves in 65 ml water, 30 ml hot water, 12.5 ml alc, 7 ml hot alc, 47 ml chloroform; slightly sol in ether. The aq soln is practically neutral.Derivative
Epicinchonine.Nomenclature
CAS number: 485-70-1
(9R)-Cinchonan-9-ol. Properties
mp 83°, [α]D22 +120.3° (c = 0.806 in alc), Rabe et al., Ann. 492, 253 (1932).Therapeutic Category
Antimalarial.
Keywords
Antimalarial