2288. Cinchophen
Nomenclature
CAS number: 132-60-5
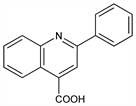
2-Phenyl-4-quinolinecarboxylic acid; 2-phenylcinchoninic acid. C16H11NO2; mol wt 249.26.
C 77.10%, H 4.45%, N 5.62%, O 12.84%.
Description and references
Quinoline derivative formerly used in treatment of chronic gout: A. B. Gutman, Arthritis Rheum. 16, 431 (1973). Prepd by heating pyruvic acid with aniline and benzaldehyde or with benzylidene aniline in abs alcohol: Doebner, Gieseke, Ann. 242, 290 (1887). Also from acetophenone and isatinic acid in alcoholic KOH: Pfitzinger, J. Prakt. Chem. 38, 582 (1882); 56, 293 (1897). Cinchophen as ulcerogenic agent: T. P. Churchill, F. H. van Wagoner, Arch. Pathol. 13, 850 (1932); N. Umeda et al., Toxicol. Appl. Pharmacol. 18, 102 (1971); T. H. Stewart et al., J. Pathol. 131, 363 (1980). Mitochondrial toxicity of cinchophen and derivatives: H. Vainio et al., Biochem. Pharmacol. 20, 1589 (1971).
Properties
Needles, mp 213-216°. Stable to air, but turns yellow under the influence of light. Slightly bitter taste. Practically insol in water. One gram dissolves in about 400 ml chloroform, in about 100 ml ether and in about 120 ml alcohol.Derivative
Hydrochloride.Nomenclature
CAS number: 132-58-1
C16H11NO2.HCl; mol wt 285.72.
C 67.26%, H 4.23%, N 4.90%, O 11.20%, Cl 12.41%.
Properties
Yellow cryst powder, slightly bitter, astringent taste; mp about 223°. Practically insol in water. Slightly sol in alcohol; sol in chloroform, ether.Derivative
Allyl ester.Nomenclature
CAS number: 524-34-5
Allyl 2-phenylcinchoninate. C19H15NO2; mol wt 289.33.
C 78.87%, H 5.23%, N 4.84%, O 11.06%.
Description and references
Prepn: Gams, US 1336952 (1920).