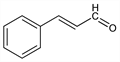
2297. Cinnamaldehyde
Nomenclature
Description and references
Found in Ceylon and Chinese cinnamon oils as the trans-form. Prepn by condensation of benzaldehyde and acetaldehyde: Peine, Ber. 17, 2117 (1884); JP 163097 (1944 to Ogawa Chem. Ind.); from 2-chloroallylbenzene: Bert, Dorier, Compt. Rend. 191, 332 (1930); Bert, Annequin, ibid. 192, 1315 (1931); by condensation of styrene with formylmethylaniline: GB 504125 (1939 to I. G. Farben); by oxidation of cinnamyl alc: Holum, J. Org. Chem. 26, 4814 (1961); Traynelis, Hergenrother, J. Am. Chem. Soc. 86, 298 (1964). Isoln from woodrotting fungus, Stereum subpileatum Berk. & Curt.: Birkinshaw et al., Biochem. J. 66, 188 (1957). Toxicity data: P. M. Jenner et al., Food Cosmet. Toxicol. 2, 327 (1964). Review of risk assessment as fragrance ingredient: D. Bickers et al., Food Chem. Toxicol. 43, 799-836 (2005); of toxicology: J. Cocchiara et al., ibid. 867-923.