Nomenclature
CAS number: 621-82-9
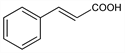
3-Phenyl-2-propenoic acid; β-phenylacrylic acid.
C
9H
8O
2; mol wt 148.16.
C 72.96%, H 5.44%, O 21.60%.
Description and references
Occurs as the trans-form in storax,
balsam Peru or Tolu, oil of cinnamon, coca leaves. Isoln: Beilstein,
Kuhlberg, Ann. 163, 123 (1872); von Miller, Ann. 188, 196 (1877). Synthesis (Perkin reaction) from benzaldehyde,
acetic anhydride, and potassium acetate: Org. React. I, 248 (1942); from oxalyl bromide
+ styrene: Treibs et al., Naturwissenschaften 45, 85 (1958); from acetylene + benzaldehyde: Herbetz, Ber. 92, 541 (1959). Prepn of cis- and trans-isomers: Comte et al., Compt. Rend. 245, 1144 (1957). Isoln from wood-rotting fungus, Stereum subpileatum Berk. & Curt.: Birkinshaw et al., Biochem. J. 66, 188 (1957). Review
of risk assessment as fragrance ingredient: D. Bickers et al., Food Chem. Toxicol. 43, 799-836 (2005); of toxicology: C. S. Letizia et al., ibid. 925-943.
Properties
Monoclinic crystals, honey, floral odor, d44 1.2475. mp 134°; bp3 147°.
Flash point, closed cup: >93.3°C. Distilling
at 146° causes decarboxylation to styrene. pK (25°) 4.46. uv max (alc):
273 nm. One gram
dissolves in about 2000 ml water at 25° (more sol in hot water), in
6 ml alc, 5 ml methanol, 12 ml chloroform. Freely sol in benzene,
ether, acetone, glacial acetic acid, carbon disulfide, oils. The
alkali salts are sol in water. LD50 (g/kg): 3.57 orally in rats; >5.0 dermally in rabbits (Letizia).Derivative
Methyl ester.
Nomenclature
CAS number: 103-26-4
C
10H
10O
2; mol wt 162.19.
C 74.05%, H 6.21%, O 19.73%.
Properties
Crystals, odor fruity and balsamic, reminiscent
of strawberries. mp 36°. d036 1.042. bp 261.9°; bp15 132.5-134°. nD21 1.5766. Freely sol in alcohol, ether.
Practically insol in water. Clearly sol in 80% alc.Derivative
Ethyl ester.
Nomenclature
CAS number: 103-36-6
Ethyl cinnamate; ethyl phenylacrylate.
C
11H
12O
2; mol wt 176.21.
C 74.98%, H 6.86%, O 18.16%.
Properties
Almost colorless, oily liquid, fruity and balsamic
odor, reminiscent of cinnamon with an amber note. d2525 1.045-1.048. d420 1.049. bp 271°. mp 6-10°. nD20 1.559-1.561. Miscible with alcohol, ether.
Insol in water. Soluble in 3 vols of 70% alc.Derivative
n-Butyl ester.
Nomenclature
CAS number: 538-65-8
Butyl cinnamate; Eliminoxy.
C
13H
16O
2; mol wt 204.26.
C 76.44%, H 7.90%, O 15.67%.
Properties
Liquid, agreeable ethereal odor when pure. bp13 145°.
d418 1.012. Very sparingly sol in water (<0.5%). Sol in 95% alc, ether,
acetone, chloroform, benzene. Incompatible with alkalies; stable
to light, air, and storage temps.Derivative
Anhydride.
Nomenclature
CAS number: 538-56-7
Cinnamic anhydride.
C
18H
14O
3; mol wt 278.30.
C 77.68%, H 5.07%, O 17.25%.
Properties
Crystals, mp 136°. Practically insol in water. Freely sol in warm
benzene; slightly sol in alc.Use
Fragrance and flavoring ingredient.
