2314. Ciprofloxacin
Nomenclature
Description and references
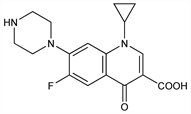
Fluorinated quinolone antibacterial. Prepn: K. Grohe et al., DE 3142854; eidem, US 4670444 (1983, 1987 both to Bayer AG); K. Grohe, H. Heitzer, Ann. 1987, 29. Antibacterial spectrum in vitro: B. Watt, F. V. Brown, J. Antimicrob. Chemother. 17, 605 (1986); C. M. Bassey et al., ibid. 623. HPLC determn in biological fluids: W. Gau et al., J. Liq. Chromatogr. 8, 485 (1985). Pharmacokinetics: G. Hoffken et al., Antimicrob. Agents Chemother. 27, 375 (1985); of extended release formulation: C. B. Washington et al., J. Clin. Pharmacol. 45, 1236 (2005). Clinical trials: C. A. Ramirez et al., ibid. 28, 128 (1985); B. E. Scully et al., Lancet 1, 819 (1986). Symposia on antibacterial spectrum and clinical use: Am. J. Med. 82, Suppl. 4A, 1-404 (1987); J. Antimicrob. Chemother. 26, Suppl. F, 3-193 (1990). Review of clinical safety and efficacy in children: R. Kubin, Infection 21, 413-421 (1993); of US FDA approval in postexposure inhalational anthrax: A. Myerhoff et al., Clin. Infect. Dis. 29, 303-308 (2004); of clinical experience in urinary tract infections: A. D. Hickerson, C. C. Carson, Expert Opin. Invest. Drugs 15, 519-532 (2006); of therapeutic potential in a broad spectrum of infections: H. Koch et al., Clin. Drug Invest. 26, 645-654 (2006).
Properties
Faintly yellowish to light yellow crystalline powder. Dec 255-257°. Sol in dilute (0.1N) HCl. Practically insol in water and ethanol.Derivative
Monohydrochloride monohydrate.Nomenclature
Properties
Light yellow crystalline powder. mp 318-320°. Slightly sol in water.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Synthetic); Quinolones and Analogs