2326. Citric Acid
Nomenclature
Description and references
Widely distributed in plants and in animal tissues and fluids. Produced by mycological fermentation on an industrial scale using crude sugar solns, such as molasses and strains of Aspergillus niger: See review by Von Loesecke, Chem. Eng. News 23, 1952 (1945); Schweiger, US 2970084 (1961 to Miles Labs.); Faith, Keyes & Clark's Industrial Chemicals, F. A. Lowenheim, M. K. Moran, Eds. (Wiley-Interscience, New York, 4th ed., 1975) pp 275-279. Also extracted from citrus fruits (lemon juice contains 5 to 8%) and from pineapple waste. Reviews: Wilson, Chem. Metall. Eng. 29, 787 (1923); Browne, Ind. Eng. Chem. 13, 81 (1921); Warneford, Hardy, ibid. 17, 1283 (1925); E. F. Bouchard, E. G. Merritt in Kirk-Othmer Encyclopedia of Chemical Technology vol. 6 (Wiley-Interscience, New York, 3rd ed., 1979) pp 150-179. Toxicity: C. M. Gruber, Jr., W. A. Halbeisen, J. Pharmacol. Exp. Ther. 94, 65 (1948).
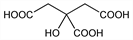
Properties
Anhydr form, mp 153°. Crystals are monoclinic holohedra and crystallize from hot concd aq soln. d 1.665. At 25°, pK1 3.128; pK2 4.761; pK3 6.396, Bates, Pinching, J. Am. Chem. Soc. 71, 1274 (1949). Soly in water: 54.0% w/w at 10°; 59.2% at 20°; 64.3% at 30°; 68.6% at 40°; 70.9% at 50°; 73.5% at 60°; 76.2% at 70°; 78.8% at 80°; 81.4% at 90°; 84.0% at 100°. LD50 in mice, rats (mmol/kg): 5.0, 4.6 i.p. (Gruber, Halbeisen).Derivative
Monohydrate.Nomenclature
Properties
Orthorhombic crystals from cold aq solns. Pleasant, sour taste. d 1.542. Monohydrate crystals lose water of crystn in dry air or when heated at about 40 to 50°, slightly deliquescent in moist air. Softens at 75°. mp ≈100°. pH of 0.1N soln = 2.2. Densities of aqueous soln (15°/15°): 10% = 1.0392; 20% = 1.0805; 30% = 1.1244; 40% = 1.1709; 50% = 1.2204; 60% = 1.2738. Soly in g/100 g satd soln: ether 2.17; chloroform 0.007; amyl alcohol 15.43; amyl acetate 5.98; ethyl acetate 5.28. Soly at 19° in g/100 g solvent: methanol 197; propanol 62.8.Derivative
Barium salt heptahydrate.Nomenclature
Properties
Powder. Loses all H2O at 150°. Sol in 1750 parts water; freely sol in dil HCl or HNO3; practically insol in alcohol.Derivative
Ethyl ester.Nomenclature
Properties
Bitter, oily liq. d20 1.137. bp760 294°; bp1.0 127°. Viscosity at 25°: 35.2 cP. Pour pt ≈10°. nD20 1.4455. Soly: water ≈6.9%; peanut oil 0.8%. Misc with alc, ether.Derivative
Disodium salt.Nomenclature
Properties
White powder, saline taste. One gram of sesquihydrate dissolves in slightly less than 2 ml water; pH of a 3% w/v soln in water: 4.9 to 5.2. LD50 in mice, rats (mmol/kg): 7.5, 7.3 i.p. (Gruber, Halbeisen).Derivative
Trisodium salt.Description and references
See Sodium Citrate.