2373. Clofazimine
Nomenclature
Description and references
Prepn: V. C. Barry et al., US 2948726 (1960 to Geigy); Barry et al., Nature 179, 1013 (1957); Barry et al., J. Chem. Soc. 1958, 859; Belton et al., Proc. R. Irish Acad. Sect. B 62, 9 (1961), C.A. 58, 4556d (1963). Activity studies: Vischer, Arzneim.-Forsch. 18, 1529 (1968). HPLC determn in plasma: J. H. Peters et al., J. Chromatogr. 229, 503 (1982). Toxicity data: Stenger et al., Arzneim.-Forsch. 20, 794 (1970). Clinical studies: Mathies, ibid. 1838; Karat et al., Br. Med. J. 1, 198 (1970). Review of clinical pharmacokinetics: M. R. Holdiness, Clin. Pharmacokinet. 16, 74-85 (1989); of clinical use: J. C. Garrelts, DICP Ann. Pharmacother. 25, 525-531 (1991). Comprehensive description: V. K. Kapoor, Anal. Profiles Drug Subs. 18, 91-120 (1989); C. M. O'Driscoll, O. I. Corrigan, Anal. Profiles Drug Subs. Excip. 21, 75-108 (1992).
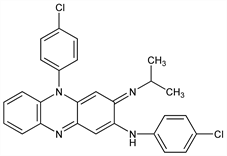
Properties
Dark-red crystals, mp 210-212°. uv max (0.01M methanolic HCl): 284, 486 nm (abs. ≈1.30, ≈0.64). pKa 8.37; also reported as 8.51. Sol in dil acetic acid, DMF. Sol in 15 parts of chloroform, 700 parts of ethanol, 1000 parts of ether. Practically insol in water. Log P (octanol/water): 7.48; (isooctane/pH 5.15 buffer) at 20°: 5.01; (n-octanol/pH 5.15 buffer) at 20°: 4.30; (n-octanol/pH 5.15 buffer) at 37°: 4.40; (n-octanol/pH 5.15 buffer) at 45°: 4.48; (n-octanol/pH 5.15 buffer) at 55°: 4.54. LD50 orally in mice, rats, and guinea pigs: >4 g/kg (Stenger).Therapeutic Category
Antibacterial (tuberculostatic, leprostatic).
Keywords
Antibacterial (Leprostatic); Antibacterial (Tuberculostatic)