2372. Clofarabine
Nomenclature
Description and references
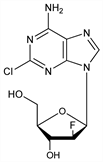
Second generation purine nucleoside analog; antimetabolite that inhibits DNA synthesis and resists deamination by adenosine deaminase. Prepn: K. A. Watanabe et al., EP 219829; eidem, US 4918179 (1987, 1990 both to Sloan-Kettering Inst. Cancer Res.); J. A. Montgomery et al., J. Med. Chem. 35, 397 (1992). Improved synthesis: W. E. Bauta et al., Org. Process Res. Dev. 8, 889 (2004). Antineoplastic activity: W. R. Waud et al., Nucleosides Nucleotides Nucleic Acids 19, 447 (2000). Clinical pharmacology: H. M. Kantarjian et al,. J. Clin. Oncol. 21, 1167 (2003). Clinical pharmacokinetics: V. Gandhi et al., Clin. Cancer Res. 9, 6335 (2003). Clinical evaluation in acute leukemia: H. Kantarjian et al., Blood 102, 2379 (2003); in pediatric patients: S. Jeha et al., ibid. 103, 784 (2004). Review of development: A. Sternberg, Curr. Opin. Investig. Drugs 4, 1479-1487 (2003); of mechanism of action and clinical experience: S. Faderl et al., Cancer 103, 1985-1995 (2005).
Properties
Crystals from water, mp 225-227° (Montgomery). Also reported as crystals from methanol, mp 237° (Bauta). uv max (water): 212, 263 nm (ε 22500, 15989).Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Antimetabolites; Purine Analogs