2385. Clometocillin
Nomenclature
CAS number: 1926-49-4
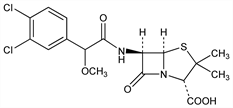
(2S,5R,6R)-6-[[(3,4-Dichlorophenyl)methoxyacetyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; 3,4-dichloro-α-methoxybenzylpenicillin; 6-(α-methoxy-3,4-dichlorophenylacetamido)penicillanic acid; clometacillin; clomethacillin; no. 356; penicillin 356. C17H18Cl2N2O5S; mol wt 433.31.
C 47.12%, H 4.19%, Cl 16.36%, N 6.46%, O 18.46%, S 7.40%.
Description and references
Semi-synthetic antibiotic related to penicillin. Prepn: Vanderhaeghe et al., Antimicrob. Agents Chemother. 1961, 581; US 3007920 (1961 to Recherche et Ind. Therap.). Activity studies: van Dijck et al., Antibiot. Chemother. 12, 192 (1962).
Derivative
Sodium salt.Nomenclature
CAS number: 54530-86-8
Rixapen (RIT). C17H17Cl2N2NaO5S; mol wt 455.29.
C 44.85%, H 3.76%, Cl 15.57%, N 6.15%, Na 5.05%, O 17.57%, S
7.04%.
Properties
[α]D +210-220°. Pure epimers were prepared by fractional crystn. First crop: Crystals, dec 232-235°. [α]D20 +182° (water). Second crop: [α]D20 +207°. Third crop: Crystals, dec 180-183°. [α]D20 +261° (water). The free acid yields two diastereoisomers: [α]D +177° and [α]D +257°.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins