2386. Clomiphene
Nomenclature
Description and references
Synthetic estrogen agonist-antagonist. Prepn: R. E. Allen et al., US 2914563 (1959 to Merrell); F. P. Palopoli et al., J. Med. Chem. 10, 84 (1967). Stereochemistry of the geometric isomers: Ernst et al., J. Pharm. Sci. 65, 148 (1976). Induction of ovulation: R. B. Greenblatt, Fertil. Steril. 12, 402 (1961). Clinical trial in anovulatory women: J. Garcia et al., ibid. 28, 707 (1977). Use in male infertility: P. J. Sorbie, R. Perez-Marrero, J. Urol. 131, 425 (1984). HPLC determn of isomers in human plasma: C. L. Baustian, T. J. Mikkelson, J. Pharm. Biomed. Anal. 4, 237 (1986). Review of development, pharmacology and clinical experience: R. P. Dickey, D. E. Holtkamp, Hum. Reprod. Update 2, 483-506 (1996).
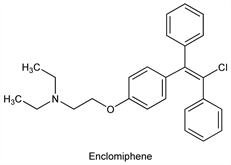
Derivative
Citrate.Nomenclature
Properties
Crystals, mp 116.5-118°. Slightly sol in water, chloroform; freely sol in methanol; sparingly sol in alcohol. Insol in ether.Derivative
cis-Form.Nomenclature
Properties
Hydrochloride salt mp 156.5-158.0°. uv max (methanol): 230, 291 nm (ε 20500, 12700).Derivative
trans-Form.Nomenclature
Properties
Hydrochloride salt mp 149.0-150.5°. uv max (methanol): 239, 297 nm (ε 22100, 11600).Therapeutic Category
Gonad-stimulating principle.
Keywords
Gonad-Stimulating Principle