2394. Clopenthixol
Nomenclature
CAS number: 982-24-1
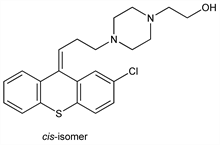
4-[3-(2-Chloro-9H-thioxanthen-9-ylidene)propyl]-1-piperazineethanol; 2-chloro-9-[3-[4-(2-hydroxyethyl)-1-piperazinyl]propylidene]thiaxanthene. C22H25ClN2OS; mol wt 400.96.
C 65.90%, H 6.28%, Cl 8.84%, N 6.99%, O 3.99%, S 8.00%.
Description and references
Thioxanthene neuroleptic. Prepn (configuration not specified): BE 585338; P. V. Petersen et al., US 3116291 (1960, 1963 both to Kefalas A/S). Prepn of the pharmacologically active cis-isomer: BE 816855; N. Lassen, US 3996211 (1974, 1976 both to Kefalas A/S). Pharmacology: Cazzullo, Andreola, Acta Neurol. 20, 162 (1965); Weissman, Mod. Probl. Pharmacopsychiatry 2, 15 (1969); Moeller Nielsen, ibid. 23. Metabolism: Khan, Acta Pharmacol. Toxicol. 27, 202 (1969). HPLC determn of isomers in serum: T. Aaes-Jorgensen, J. Chromatogr. 188, 239 (1980). Series of articles on pharmacology and clinical studies: Acta Psychiatr. Scand. 64, Suppl. 294, 1-77 (1981).
Properties
Colorless syrup. Sparingly sol in ether. Readily sol in methanol.Derivative
Dihydrochloride.Nomenclature
CAS number: 633-59-0
AY-62021; N-746; Ciatyl (Troponwerke); Sordenac (Lundbeck); Sordinol (Ayerst). C22H25ClN2OS.2HCl; mol wt 473.89.
C 55.76%, H 5.74%, Cl 22.44%, N 5.91%, O 3.38%, S 6.77%.
Properties
Crystals from ethanol, mp 250-260° (dec). Freely sol in water; sparingly sol in alcohol. Practically insol in other organic solvents. LD50 in male mice (mg/kg): 111 i.v. (Lassen).Derivative
cis(Z)-Form.Nomenclature
CAS number: 53772-83-1
α-Clopenthixol; zuclopenthixol. Properties
Crystals, mp 84-85°.Derivative
cis(Z)-Form dihydrochloride.Nomenclature
CAS number: 58045-23-1
Cisordinol (Lundbeck); Clopixol (HMR). Properties
Crystals, mp 250-260° (dec). LD50 in male mice (mg/kg): 105 i.v. (Lassen).Therapeutic Category
Antipsychotic.
Keywords
Antipsychotic; Thioxanthenes