2413. Clostebol
Nomenclature
CAS number: 1093-58-9
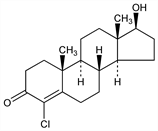
(17β)-4-Chloro-17-hydroxyandrost-4-en-3-one; 4-chlorotestosterone. C19H27ClO2; mol wt 322.87.
C 70.68%, H 8.43%, Cl 10.98%, O 9.91%.
Description and references
Prepn: Camerino et al., Farmaco Ed. Sci. 11, 586 (1956); eidem, J. Am. Chem. Soc. 78, 3540 (1956); Ringold et al., J. Org. Chem. 21, 1432 (1956); Camerino, US 2953582 (1960 to Farmitalia); Julian, Printy, US 2933510 (1960 to Julian Labs.). Clinical pharmacology of the acetate: Molinatti et al., Folia Endocrinol. 14, 528 (1961); Krueskemper, Morgner, Int. Z. Klin. Pharmakol. Ther. Toxikol. 1, 455 (1968).
Properties
Crystals from acetone + hexane, mp 188-190°. [α]D20 +148° (CHCl3). uv max (95% ethanol): 256 nm (log ε 4.13).Derivative
Acetate.Nomenclature
CAS number: 855-19-6
Macrobin; Steranabol (Farmitalia). C21H29ClO3; mol wt 364.91.
C 69.12%, H 8.01%, Cl 9.72%, O 13.15%.
Properties
Crystals from methanol, mp 228-230°. [α]D +118 ±4° (CHCl3). uv max: 255 nm (ε 13,300). Sol in ethanol.Note
This is a controlled substance (anabolic steroid): 21 CFR, 1308.13, as defined in 1300.01.Therapeutic Category
Anabolic.
Keywords
Anabolic