2419. Cloxacillin
Nomenclature
CAS number: 61-72-3
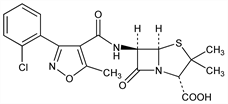
(2S,5R,6R)-6-[[[3-(2-Chlorophenyl)-5-methyl-4-isoxazolyl]carbonyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; [3-(o-chlorophenyl)-5-methyl-4-isoxazolyl]penicillin; [5-methyl-3-(o-chlorophenyl)-4-isoxazolyl]penicillin; 6-[3-(o-chlorophenyl)-5-methyl-4-isoxazolecarboxamido]penicillanic
acid. C19H18ClN3O5S; mol wt 435.88.
C 52.35%, H 4.16%, Cl 8.13%, N 9.64%, O 18.35%, S 7.36%.
Description and references
Semi-synthetic antibiotic related to penicillin; chlorinated deriv of oxacillin, q.v. Prepn: F. P. Doyle, J. H. C. Nayler, US 2996501 (1961); F. P. Doyle et al., J. Chem. Soc. 1963, 5838. Manuf: Ind. Chem. 39, 513 (1963), C.A. 60, 1543a (1964). Properties and pharmacology: J. H. C. Nayler et al., Nature 195, 1264 (1962). Toxicity data: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Comprehensive description: D. L. Mays, Anal. Profiles Drug Subs. 4, 113-136 (1975).
Derivative
Sodium salt monohydrate.Nomenclature
CAS number: 7081-44-9;
642-78-4
(anhydrous)
Sodium cloxacillin; BRL-1621; Cloxapen (GSK); Cloxypen (BMS); Ekvacillin (AstraZeneca); Latocillin (Chassot); Orbenin (GSK); Methocillin-S (Meiji); Tegopen (BMS). C19H17ClN3NaO5S.H2O; mol wt 475.88.
C 47.95%, H 4.02%, Cl 7.45%, N 8.83%, Na 4.83%, O 20.17%, S
6.74%.
Properties
Microcryst powder, dec 170°. [α]D20 +163° (c = 1 in water). pH of 1% aq soln = 6.0-7.5. Sol in water, methanol, ethanol, pyridine, ethylene glycol. LD50 in rats, mice (mg/kg): 1630 ±112, 1280 ±50 i.p. (Goldenthal).Derivative
Benzathine salt.Nomenclature
CAS number: 23736-58-5
Dry Clox (Wyeth); Noroclox (Norbrook). C54H56Cl2N8O10S2; mol wt 1112.11.
C 58.32%, H 5.08%, Cl 6.38%, N 10.08%, O 14.39%, S 5.77%.
Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins