2451. Cobamamide
Nomenclature
Description and references
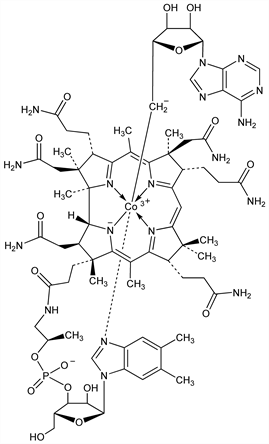
One of the metabolically active forms of vitamin B12; characterized by a 5′-deoxyadenosyl nucleoside linked by a C to Co bond. Cofactor for several bacterial enzymes that catalyze carbon-skeleton rearrangements, heteroatom eliminations, and amino group migrations. Required by mammals as coenzyme for methylmalonyl-CoA mutase in the catabolism of certain amino acids and fatty acids. Discovery: H. A. Barker et al., Proc. Natl. Acad. Sci. USA 44, 1093 (1958) DOI PubMed. Isoln from Clostridium tetanomorphum: idem et al., J. Biol. Chem. 235, 181 (1960) PubMed; from Propionibacterium shermanii: eidem, ibid. 480 PubMed. Prepn from a cobalamin-thiol complex: M. Murakami et al., US 3461114 (1969 to Yamanouchi). Crystal structure: P. G. Lenhert, D. C. Hodgkin, Nature 192, 937 (1961) DOI PubMed. Review of uptake and metabolism by humans and role in deficiency diseases: D. Watkins, D. S. Rosenblatt, Endocrinologist 11, 98-104 (2001). Review of biosynthesis by bacteria: M. J. Warren et al., Nat. Prod. Rep. 19, 390-412 (2002) DOI PubMed; of mechanisms of B12-dependent reactions: R. Banerjee, S. W. Ragsdale, Annu. Rev. Biochem. 72, 209-247 (2003) DOI PubMed; R. Banerjee, Chem. Rev. 103, 2083-2094 (2003) DOI PubMed; T. Toraya, ibid. 2095-2127 (2003) DOI PubMed.
Properties
Yellow-orange six-faced crystals which become deep red upon exposure to air. Absorption max (H2O): 260, 375, 522 nm (A × 10-6 34.7, 10.9, 8.0 cm2/mole). Soly in water (24°): 16.4 mmol. Solns of pH <3.5 are yellow, >3.5, red. Sol in ethanol, phenol. Practically insol in acetone, ether, dichloroethylene, dioxane. pKa 3.5. Stability studies: Collado, Nieto, Ann. Pharm. Fr. 27, 427 (1969) PubMed. Highly sensitive to light, to cyanide and moderately sensitive to acid. Solns are most stable at pH 6-7 stored in the dark. Heating of acid or alkaline solns produces slow inactivation.Therapeutic Category
Enzyme cofactor; vitamin (hematopoietic).
Therapeutic Category (Veterinary)
Vitamin (hematopoietic).Keywords
Vitamin/Vitamin Source; Vitamin B12