10014. Vitamin B12
Nomenclature
Description and references
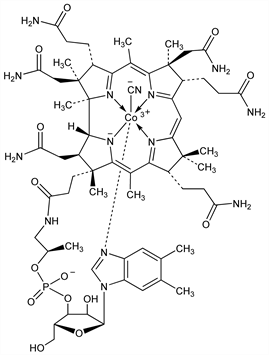
Prototype of the family of naturally occurring cobalt coordination compds known as corrinoids. Analogs of vitamin B12 which differ only in the β-ligand of the cobalt are termed cobalamins. Synthesized almost exclusively by bacteria. Dietary sources include fish, meat, liver, and dairy products; plants have little or no cobalamins. Also found in soil and water, the richest sources being activated sewage sludge (see Milorganite) or manure. Converted by the body into its bioactive forms, methylcobalamin and cobamamide, q.q.v., which serve as enzyme cofactors. Severe deficiency may result in megaloblastic anemia and/or neurological impairment. Isoln from mammalian liver: E. L. Rickes et al., Science 107, 396 (1948) DOI PubMed; from cultures of Streptomyces griseus: eidem, ibid. 108, 634 (1948) DOI PubMed; E. L. Rickes, T. R. Wood, US 2563794 (1951 Merck & Co.). Fermentation process using Pseudomonas denitrificans: R. A. Long, US 3018225 (1962 to Merck & Co.). Purification from sewage sludge: P. J. Van Melle, US 3057851 (1962 to Armour). Structure: D. C. Hodgkin et al., Nature 176, 325 (1955) DOI PubMed; R. Bonnett et al., ibid. 328. X-ray structure analysis: D. C. Hodgkin, Fortschr. Chem. Org. Naturst. 15, 167-227 (1958) PubMed. Stereochemistry: Stora, Bull. Soc. Chim. Fr. 1959, 1421. Total synthesis: R. B. Woodward, Pure Appl. Chem. 33, 145 (1973) DOI PubMed. Nomenclature: IUPAC rules, ibid. 48, 497 (1976). Gastrointestinal absorption is dependent on Castle's intrinsic factor, q.v., and haptocorrin (R-protein); transport of the absorbed vitamin is mediated by transcobalamins: B. Seetharam, D. H. Alpers, Annu. Rev. Nutr. 2, 343-369 (1982) DOI PubMed; R. Grsbeck, Clin. Biochem. 17, 99-107 (1984) DOI PubMed. Role of Co‐C bond in B12-dependent reactions: J. M. Pratt, Pure Appl. Chem. 65, 1513 (1993) DOI. Review of biosynthesis: A. I. Scott, Angew. Chem. Int. Ed. 32, 1223-1243 (1993) DOI. Comprehensive description: J. Kirschbaum, Anal. Profiles Drug Subs. 10, 183-288 (1981). Book: B12 vols. 1 and 2, D. Dolphin, Ed. (Wiley-Interscience, New York, 1982) 672 and 506 pp. Reviews: B. T. Golding in Comprehensive Organic Chemistry vol. 5, E. Haslam, Ed. (Pergamon, New York, 1979) pp 549-584; “Vitamin B12” in Vitamins, W. Friedrich, Ed. (de Gruyter, Berlin, 1988) pp 837-928. Review of chemistry and enzymology: K. L. Brown, Chem. Rev. 105, 2075-2149 (2005) DOI PubMed.
Properties
Hygroscopic, dark red crystals. When exposed to air, may absorb ≈12% water. The hydrated crystals are stable to air. Darkens at 210-220°. Not melted at 300°. [α]23656 59 ± 9° (dil aq soln). Absorption max (water): 278, 361, 550 nm (A1%1cm 115, 204, 64). Odorless and tasteless. One gram dissolves in ≈80 ml water. Aq solns are neutral, maximum stability in the pH range 4.5-5. Solns in this pH range can be autoclaved for 20 min at 120°. Soluble in alc. Insol in acetone, CHCl3, ether.Therapeutic Category
Vitamin (hematopoietic).
Therapeutic Category (Veterinary)
Vitamin (hematopoietic).Keywords
Vitamin/Vitamin Source; Vitamin B12