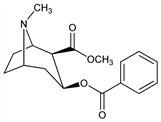
2455. Cocaine
Nomenclature
Description and references
From the leaves of Erythroxylon coca Lam. and other species of Erythroxylon, Erythroxylaceae or by synthesis. Extraction procedure: Squibb, Pharm. J. [3] 15, 775, 796; 16, 67 (1885); Emde in Ullmanns Encyklop"adie der technischen Chemie Schwyzer, Die Fabrikation pharmazeutischer und chemisch-technischer Produkte (Berlin, 1931). Configuration: Findlay, J. Am. Chem. Soc. 76, 2855 (1954); O. Kovacs et al., Helv. Chim. Acta 37, 892 (1954). Synthesis: R. Willst"atter et al., Ann. 434, 111 (1923). Stereospecific synthesis of dl-form: J. J. Tufariello et al., Tetrahedron Lett. 1978, 1733; eidem, J. Am. Chem. Soc. 101, 2435 (1979). Biosynthesis: E. Leete, S. H. Kim, J. Am. Chem. Soc. 110, 2976 (1988). Absorption spectrum: J. J. Dobbie, J. J. Fox, J. Chem. Soc. 103, 1193 (1913); Fischer, Arch. Exp. Pathol. Pharmakol. 170, 610 (1933). Toxicity: C. L. Rose et al., J. Lab. Clin. Med. 15, 731 (1930). Vapor pressure studies: A. H. Lawrence et al., Can. J. Chem. 62, 1886 (1984). Comprehensive description: F. J. Muhtadi, A. A. Al-Badr, Anal. Profiles Drug Subs. 15, 151-231 (1986). Review of pharmacology and pharmacotherapies for abuse: M. Rocío et al., Bioorg. Med. Chem. 12, 5019-5030 (2004).
Properties
Monoclinic tablets from alcohol, mp 98°. Volatile, esp above 90°, but the sublimate is not crystalline. bp0.1 187-188°. [α]D18 -35° (50% alcohol); [α]D20 -16° (c = 4 in chloroform). Aq solns are alkaline to litmus. pKa (15°) 8.61. pKb (15°) 5.59. One gram dissolves in 600 ml water, 270 ml water at 80°, 6.5 ml alcohol, 0.7 ml chloroform, 3.5 ml ether, 12 ml oil turpentine, 12 ml olive oil, 30-50 ml liquid petrolatum. Also sol in acetone, ethyl acetate, carbon disulfide. LD50 i.v. in rats: 17.5 mg/kg (Rose).Derivative
Hydrochloride.Nomenclature
Properties
Crystals, granules, or powder; saline, slightly bitter taste; numbs tongue and lips. mp ≈195°. [α]D -72° (c = 2 in aq soln pH 4.5). One gram dissolves in 0.4 ml water; 3.2 ml cold, 2 ml hot alcohol; 12.5 ml chloroform. Also sol in glycerol, acetone. Insol in ether or oils. Avoid heat in preparing soln as it decomposes. Preserve in well-closed, light-resistant containers.Note
This is a controlled substance: 21 CFR, 1308.12.Therapeutic Category
Anesthetic (local).
Therapeutic Category (Veterinary)
Topical anesthetic (ophthalmic).Keywords
Anesthetic (Local)