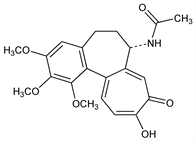
2470. Colchiceine
Nomenclature
CAS number: 477-27-0
N-[(7S)-5,6,7,9-Tetrahydro-10-hydroxy-1,2,3-trimethoxy-9-oxobenzo[a]heptalen-7-yl]acetamide; 7-acetamido-10-hydroxy-1,2,3-trimethoxy-6,7-dihydrobenzo[a]heptalen-9(5H)-one. C21H23NO6; mol wt 385.41.
C 65.44%, H 6.02%, N 3.63%, O 24.91%.
Description and references
Deriv of colchicine, q.v., isolated from Colchicum autumnale L., Liliaceae: Santavy, Macák, Collect. Czech. Chem. Commun. 19, 805 (1954). Toxicity data: B. Goldberg et al., Cancer 3, 124 (1950). Structure and synthesis: Nakamura, Chem. Pharm. Bull. 10, 299 (1962). Circular dichroism: J. Hrbek et al., Collect. Czech. Chem. Commun. 47, 2258 (1982). Effect on antibody response induced in vitro: J. Sterzl et al., Folia Microbiol. 27, 256 (1982).
Properties
Yellow crystals from dioxane + ether, mp 178-179°. [α]D25 -255.1° (c = 1 in chloroform). uv max (95% ethanol): 351, 244 nm (log ε 4.28, 4.51). Slightly sol in water; freely sol in alcohol, chloroform. Almost insol in benzene, ether.Derivative
Ethyl ether.C23H27NO6; mol wt 413.46.
C 66.81%, H 6.58%, N 3.39%, O 23.22%.