2471. Colchicine
Nomenclature
Description and references
Principal alkaloid of the meadow saffron, Colchicum autumnale L., Liliaceae. Binds to tubulin and inhibits microtubule polymerization. Isoln: P. J. Pelletier, J. B. Caventou, Ann. Chim. Phys. [2] 14, 69 (1820). Extraction procedure from seeds: F. Chemnitius, J. Prakt. Chem. [II] 118, 29 (1928) DOI; F. Santavy, T. Reichstein, Helv. Chim. Acta 33, 1606 (1950) DOI; by supercritical CO2: E. Ellington et al., Phytochem. Anal. 14, 164 (2003) DOI PubMed. Proposed structure: A. Windaus, Ann. 439, 58 (1924); M. J. S. Dewar, Nature 155, 141 (1945). Confirmation of structure: M. V. King et al., Acta Crystallogr. 5, 437 (1952) DOI. Abs config: H. Corrodi, E. Hardegger, Helv. Chim. Acta 38, 2030 (1955) DOI. Crystal structure: L. Lessinger, T. N. Margulis, Acta Crystallogr. B34, 578 (1978) DOI. Total synthesis of (±)-form: J. Schreiber et al., Helv. Chim. Acta 44, 540 (1961) DOI; E. E. van Tamelen et al., Tetrahedron 14, 8 (1961) DOI. Biosynthesis: E. Leete, Tetrahedron Lett. 6, 333 (1965) DOI; A. R. Battersby et al., J. Chem. Soc. 1964, 4257 DOI. Toxicity: S. J. Rosenbloom, F. C. Ferguson, Toxicol. Appl. Pharmacol. 13, 50 (1968) DOI; R. P. Beliles, ibid. 23, 537 (1972) DOI PubMed. Comprehensive description: D. K. Wyatt et al., Anal. Profiles Drug Subs. 10, 139-182 (1981). Review of syntheses: T. Graening, H.-G. Schmalz, Angew. Chem. Int. Ed. 43, 3230-3256 (2004) DOI. Review of interaction with tubulin: B. Bhattacharyya et al., Med. Res. Rev. 28, 155-183 (2008) DOI PubMed; of pharmacology and clinical experience: C. Cerquaglia et al., Curr. Drug Targets Inflamm. Allergy 4, 117-124 (2005) DOI PubMed; G. Nuki, Curr. Rheumatol. Rep. 10, 218-227 (2008) DOI PubMed.
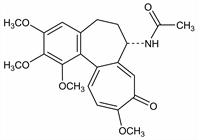
Properties
Nearly colorless to pale yellow needles from ethyl acetate + ether, mp 154-156°. Darkens on exposure to light. [α]D22 -121.6° (c = 1.001 in chloroform). pK at 20°: 12.35. pH of 0.5% soln: 5.9. uv max (ethanol): 247, 350 nm (log ε 4.45; 4.20). One gram dissolves in 22 ml water, 220 ml ether, 100 ml benzene; freely sol in alcohol or chloroform. Practically insol in petr ether. LD50 in rats (mg/kg): 1.6 i.v. (Rosenbloom, Ferguson); in mice (mg/kg): 4.13 i.v. (Beliles).Use
In plant genetics research (for doubling chromosomes).Therapeutic Category
Gout suppressant. In treatment of familial Mediterranean fever.
Keywords
Antigout