2477. Colfosceril Palmitate
Nomenclature
Description and references
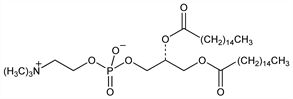
Major phospholipid constituent and primary surface tension lowering component of natural lung surfactant. Isoln from lung extracts: S. J. Thannhauser et al., J. Biol. Chem. 166, 669 (1946); E. S. Brown, Am. J. Physiol. 207, 402 (1964). Enantioselective synthesis: E. Baer, M. Kates, J. Am. Chem. Soc. 72, 942 (1950). Clinical study in combination therapy for neonatal respiratory distress syndrome (RDS): C. J. Morley et al., Lancet 1, 64 (1981). Diagnostic use as predictor of fetal lung maturity: A. Lohninger et al., Clin. Chem. 29, 650 (1983). HPTLC determn in amniotic fluid: J. G. Alvarez et al., J. Chromatogr. B 665, 79 (1995). Review of early literature: D. F. Tierney, Am. J. Physiol. 257, L1-L12 (1989); of role in pulmonary function and clinical use: L. M. G. van Golde et al., Physiol. Rev. 68, 374-455 (1988); of structure and function as surface active agent: B. A. Hills, Br. J. Anaesth. 65, 13-29 (1990); of biosynthesis: H. P. Haagsman, L. M. G. van Golde, Annu. Rev. Physiol. 53, 441-464 (1991).
Properties
Occurs as monohydrate, crystals from hot diisobutyl ketone, softens at 75-79°, mp 234-235°. [α]D23 +6.6° (c = 4.2 in 1:1 chloroform-methanol). Readily sol in chloroform, hot diisobutyl ketone, hot dioxane. Readily emulsified in water. Soly at 22-23° (g/100 ml): ethanol 1.5, ether 0.02, acetone 0.02, pyridine 1.1, acetic acid 4.0, methanol 1.4.Therapeutic Category
Pulmonary surfactant. Diagnostic aid (fetal lung maturity).
Keywords
Diagnostic Aid; Pulmonary Surfactant