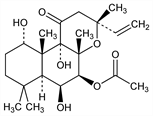
2476. Colforsin
Nomenclature
Description and references
Diterpene isolated from Coleus forskohlii, Briq. Labiatae, possessing vasodilating and cardiostimulatory properties. Isoln and characterization: S. V. Bhat et al., Tetrahedron Lett. 1977, 1669; eidem, DE 2557784; eidem, US 4088659 (1977, 1978 both to Hoechst). Synthesis by hydroxylation of 9-deoxyforskolin: N. J. Hrib, Tetrahedron Lett. 28, 19 (1987); see also F. E. Ziegler et al., J. Am. Chem. Soc. 109, 8115 (1987). Stereocontrolled synthesis of the 3-ring system: S. Hashimoto et al., Chem. Commun. 1987, 24. Total synthesis of (±)-forskolin: S. Hashimoto et al., J. Am. Chem. Soc. 110, 3670 (1988); E. J. Corey et al., ibid. 3672. Positive inotropic and blood-pressure lowering activity: E. Lindner et al., Arzneim.-Forsch. 28, 284 (1978). Activates adenylate cyclase: H. Metzger, E. Lindner, ibid. 31, 1248 (1981). Binds to catalytic site of adenylate cyclase: T. Pfeuffer, H. Metzger, FEBS Lett. 146, 369 (1982). Lowers intraocular pressure: J. Capriolli, M. Sears, Lancet 1, 958 (1983). In treatment of glaucoma: eidem, US 4476140 (1984 to Yale University). Reviews: K. B. Seaman, J. W. Daly, J. Cyclic Nucleotide Res. 1981, 201; K. B. Seaman, “Forskolin and Adenylate Cyclase: New Opportunities in Drug Design” in Annu. Rep. Med. Chem. 19, D. M. Bailey, Ed. (Academic Press, New York, 1984) pp 293-302.