2539. Cortisone
Nomenclature
Description and references
Isoln from suprarenal glands: Pfiffner et al., J. Biol. Chem. 111, 585 (1935); 116, 291 (1936); Mason et al., ibid. 114, 613 (1936); Reichstein, Helv. Chim. Acta 19, 1107 (1936); Kuizenga, Cartland, Endocrinology 24, 526 (1939). Synthesis of the monoacetate from desoxycholic acid: Sarett, J. Biol. Chem. 162, 601 (1946). Further development of prepn methods: Meystre, Wettstein, Experientia 3, 185 (1947); Helv. Chim. Acta 30, 1037, 1256 (1947); Reichstein et al., ibid. 26, 562, 705, 721 (1943); 27, 821 (1944); Reichstein, US 2403683 (1946); Kendall et al., J. Biol. Chem. 166, 345 (1946); Gallagher, US 2447325 (1948); Peterson, Murray, J. Am. Chem. Soc. 74, 1871 (1952); Perlman, ibid. 2126; Sarett, ibid. 70, 1454 (1948); 71, 2443 (1949); Mattox, Kendall, ibid. 70, 882 (1948). Stereospecific total synthesis: Sarett et al., ibid. 74, 4974 (1952).
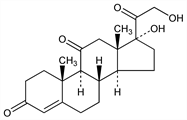
Properties
Rhombohedral platelets from 95% alcohol, mp 220-224° (some decompn) when heated in evac capillary. [α]D25 +209° (c = 1.2 in 95% alcohol); [α]25546 +269° (c = 0.125 in benzene); [α]25546 +248° (c = 0.1 to 0.2 in alcohol). uv max: 237 nm (ε 1.4×104) see Mason et al., J. Biol. Chem. 116, 267 (1936); Wintersteiner, Pfiffner, ibid. 291. Fairly sol in cold methanol, ethanol, acetone; much less sol in ether, benzene, chloroform; slightly sol in water (28 mg/100 ml at 25°). The water soln is neutral. Gives orange-red soln with intense green fluorescence in concd H2SO4. Reduces Benedict's soln on heating.Derivative
21-Acetate.Nomenclature
Properties
Flat needles from acetone; clusters of radiating rods from chloroform. Becomes opaque at 70-100°, mp 235-238° with slight sintering at 230°. [α]D25 +164° (c = 0.5 in acetone), [α]D25 +208 to +217° (dioxane). uv max: 238 nm (ε 1.58×104), see Sarett, J. Biol. Chem. 162, 630 (1946). Soly at 25° in water: 2.2 mg/100 ml; in propylene glycol 44 mg/100 ml; in chloroform 182 mg/g. Reduces ammoniacal silver nitrate soln at room temp. Sol in sulfuric acid giving a yellow soln without fluorescence (difference from hydrocortisone acetate).Derivative
21-Cyclopentanepropionate.Nomenclature
Properties
Needles from diisopropyl ether, mp 158-161°. [α]D20 +190° (chloroform). uv max (ethanol): 239 nm (ε 16350). Sol in ether, glycols, vegetable oils, especially sesame, peanut, and corn oils.Therapeutic Category
Glucocorticoid.
Therapeutic Category (Veterinary)
Glucocorticoid, anti-inflammatory agent.Keywords
Glucocorticoid