2538. Corticosterone
Nomenclature
CAS number: 50-22-6
(11β)-11,21-Dihydroxypregn-4-ene-3,20-dione; 4-pregnene-11β,21-diol-3,20-dione; Kendall's compound B; Reichstein's substance H; compd B. C21H30O4; mol wt 346.46.
C 72.80%, H 8.73%, O 18.47%.
Description and references
Endogenous glucocorticoid; intermediate in the biosynthesis of aldosterone from progesterone. Isoln from adrenal cortex: Mason et al., J. Biol. Chem. 114, 613 (1936); Reichstein, von Euw, Helv. Chim. Acta 21, 1197 (1938); Jeanloz et al., US 2676904 (1954 to Searle); from adrenal extract: Reichstein, US 2166877 (1939 to Roche-Organon). Prepn from desoxycholic acid: Wallis, Chakravorty, US 2341250 (1944 to Research Corp.); from 11-deoxycorticosterone: Zaffaroni, US 2671752 (1954 to Syntex); from cortisone: Oliveto et al., US 2927108 (1960 to Schering).
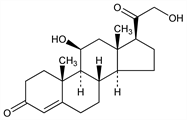
Properties
Trigonal plates from acetone, mp 180-182°. [α]D15 +223° (c = 1.1 in alc). uv max: 240 nm. Insol in water. Sol in usual organic solvents. Upon moistening with concd H2SO4 it gives an orange-yellow soln which has a strong fluorescence.Derivative
21-Acetate.C23H32O5; mol wt 388.50.
C 71.11%, H 8.30%, O 20.59%.