2547. Corynantheine
Nomenclature
CAS number: 18904-54-6
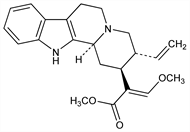
(16E)-16,17,18,19-Tetradehydro-17-methoxycorynan-16-carboxylic
acid methyl ester; (E)-16,17,18,19-tetradehydro-17-methoxy-17,18-secoyohimban-16-carboxylic
acid methyl ester. C22H26N2O3; mol wt 366.45.
C 72.11%, H 7.15%, N 7.64%, O 13.10%.
Description and references
Isolated from Corynanthe johimbe K. Schum., and Pseudocinchona africana A. Cheval., Rubiaceae: Karrer et al., Helv. Chim. Acta 9, 1059 (1926); 35, 851 (1952). Structure: Janot et al., ibid. 34, 1207 (1951). Stereochemistry: Van Tamelen et al., J. Am. Chem. Soc. 79, 6426 (1957). Total synthesis of dl- form: Van Tamelen, Wright, Tetrahedron Lett. 1964, 295; eidem, J. Am. Chem. Soc. 91, 7349 (1969). Total synthesis of d-form: Autrey, Scullard, ibid. 90, 4917 (1968).
Properties
Crystals: α-form mp 103-107°; β-form mp 165-166°. [α]D +28° (c = 1 in methanol). uv max (methanol): 227, 280, 291 nm (log ε 4.64, 3.82, 3.80).Derivative
Hydrochloride.C22H26N2O3.HCl; mol wt 402.91.
C 65.58%, H 6.75%, N 6.95%, O 11.91%, Cl 8.80%.
Properties
Needles from alcohol + ether, mp 194-206°. Also reported as mp 176-179°. [α]D20 +12° (water). Sol in alcohol, sparingly sol in water.Derivative
Dihydrocorynantheine.C22H28N2O3; mol wt 368.47.
C 71.71%, H 7.66%, N 7.60%, O 13.03%.
Properties
Total synthesis of dl-form: Van Tamelen, Hester, J. Am. Chem. Soc. 81, 3805 (1959); eidem, ibid. 91, 7342 (1969). Plates from alcohol + water, mp 177-177.5°. [α]D +28°.Derivative
Hydrochloride.C22H28N2O3.HCl; mol wt 404.93.
C 65.25%, H 7.22%, N 6.92%, O 11.85%, Cl 8.76%.