2548. Corynanthine
Nomenclature
CAS number: 483-10-3
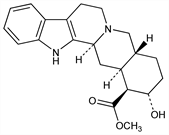
(16β,17α)-17-Hydroxyyohimban-16-carboxylic acid methyl ester; rauhimbine. C21H26N2O3; mol wt 354.44.
C 71.16%, H 7.39%, N 7.90%, O 13.54%.
Description and references
From bark of Pseudocinchona africana Chev., Corynanthe johimbe K. Schum., Rubiaceae and Rauwolfia serpentina (L.) Benth., Apocynaceae: Raymond-Hamet, Compt. Rend. 212, 305 (1941); Jorio, Ann. Chim. Farm. 1939, 50, C.A. 33, 93069 (1939); Le Hir et al., Ann. Pharm. Fr. 11, 546 (1953); Hofmann, Helv. Chim. Acta 37, 314 (1954). Identity with rauhimbine: idem, ibid. 849. Structure and stereochemistry: Janot et al., Bull. Soc. Chim. Fr. 1952, 1085; 1961, 637.
Properties
Stout prisms from acetone, dec 225-226°. [α]D19 -85° (c = 0.5 in pyridine). uv max (methanol): 226, 283, 290 nm (log ε 4.56, 3.87, 3.79). Practically insol in water or petr ether. Sol in 40 parts of boiling chloroform, in 60 parts of boiling benzene, in 20 parts of boiling ethyl acetate, in 5 parts of boiling alcohol.Derivative
O,N-Dibutyrylcorynanthine hydrochloride.Description and references
Prepn: Reiser et al., US 2975183 (1961 to Chemische Werke Albert).
Properties
Crystals from benzene, mp 208-210°.Derivative
O,N-Dipropionylcorynanthine hydrochloride.Description and references
Prepn: Reiser et al., loc. cit.