2579. Cresols
Nomenclature
Description and references
Mixture of the three isomeric cresols, in which the m-isomer predominates. Obtained from coal tar: Paulsen, US 2998457 (1962 to Ashland Oil & Ref.). Usually contains a few per cent phenol. Prepn by sulfonation of toluene: Englund et al., Ind. Eng. Chem. 45, 189 (1953); by oxidation of toluene: Braunwarth, Winsted, US 2994722 (1961 to Pure Oil). Toxicity study: W. B. Deichmann, S. Witherup, J. Pharmacol. Exp. Ther. 80, 233 (1944). Review of manuf processes: Faith, Keyes & Clark's Industrial Chemicals, F. A. Lowenheim, M. K. Moran, Eds. (Wiley-Interscience, New York, 4th ed., 1975) pp 285-293; of toxicology and human exposure: Toxicological Profile for Cresols (PB93-110732, 1992) 175 pp.
Properties
Colorless, yellowish, brownish-yellow or pinkish liq; phenolic odor; becomes darker with age and on exposure to light. Poisonous. d2525 1.030-1.038. Not less than 90% by vol distills between 195-205°. Soluble in about 50 parts water; miscible with alcohol, benzene, ether, glycerol, petr ether; also sol in solns of fixed alkali hydroxides. A soln in water is neutral to bromocresol purple. Protect from light.Derivative
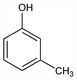
m-Cresol.Nomenclature
Description and references
Obtained from coal tar: Maesawa, Kurakano, JP 55 8929 (1955 to Osaka Gas), C.A. 52, 1231d (1958); Macak, Rehak, Brennst.-Chem. 43, 80 (1962). Prepn from toluene: Toland, US 2760991 (1956 to California Res. Corp.); by oxidation of o- or p-toluic acid: Kaeding et al., Ind. Eng. Chem. 53, 805 (1961).
Properties
Colorless or yellowish liquid; phenolic odor. d420 1.034. bp 202°. mp 11-12°. Flash pt, closed cup: 187°F (86°C). nD20 1.5398. Sol in about 40 parts water, in solns of fixed alkali hydroxides; miscible with alc, chloroform, ether. LD50 orally in rats: 2.02 g/kg (Deichmann, Witherup).Derivative
o-Cresol.Nomenclature
Description and references
Prepn from m-toluic acid: Toland, US 2766294 (1956 to California Res. Corp.); Barnard, Meyer, US 2852567 (1958 to Dow).
Properties
Crystals or liq, becoming dark with age and exposure to air and light; phenolic odor. d420 1.047. bp 191-192°. mp 30°. Flash pt 81-83°C. nD20 1.553. Sol in about 40 parts water, in solns of the fixed alkali hydroxides. Miscible with alcohol, chloroform, ether. Protect from light. LD50 orally in rats: 1.35 g/kg (Deichmann, Witherup).Derivative
p-Cresol.Nomenclature
Description and references
Obtained from coal tar. Laboratory prepn from p-toluenesulfonic acid by fusion with potassium hydroxide: W. W. Hartman, Org. Synth. coll. vol. I, 175 (2nd ed., 1941); from toluene: Braunwarth, US 3046305 (1962 to Pure Oil).
Properties
Crystals. Phenolic odor. d420 1.0341. mp 35.5°. bp760 201.8°; bp200 179.4°; bp100 140.0°; bp40 117.7°; bp20 102.3°; bp10 88.6°; bp5 76.5°; bp1.0 53.0°. Flash pt (closed cup) 86°C (187°F). Volatile in steam. nD20 1.5395. 100 ml water dissolves about 2.5 g at 50°, about 5 g at 100°. Sol in aq solns of alkali hydroxides; in the usual organic solvents. LD50 orally in rats: 1.8 g/kg (Deichmann, Witherup).Caution
Potential symptoms of overexposure to o-, m- or p-isomers are irritation of eyes, skin and mucous membranes; CNS effects; confusion, depression and respiratory failure; dyspnea, irregular rapid respiration and weak pulse; skin and eye burns; dermatitis; lung, liver, kidney and pancreas damage. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 78. Symptoms of intoxication are similar to those produced by phenol, q.v. Acute exposure may cause muscular weakness, gastroenteric disturbances, severe depression, collapse and death. Chronic exposure may cause digestive disturbances, liver and kidney damage, and skin eruptions. Cresol has a marked corrosive action on tissues, producing burns and dermatitis. See Patty's Industrial Hygiene and Toxicology vol. 2A, G. D. Clayton, F. E. Clayton, Eds. (Wiley-Interscience, New York, 3rd ed., 1981) pp 2597-2601.Use
For making synthetic resins; in disinfectants and fumigants; as industrial solvent; m-cresol in photographic developers, explosives.Therapeutic Category
Disinfectant.
Therapeutic Category (Veterinary)
Local antiseptic, parasiticide, disinfectant; has been used as an intestinal antiseptic.Keywords
Antiseptic/Disinfectant; Phenols