2621. Cuminaldehyde
Nomenclature
CAS number: 122-03-2
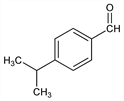
4-(1-Methylethyl)benzaldehyde; p-isopropylbenzaldehyde; cuminal; cumaldehyde. C10H12O; mol wt 148.20.
C 81.04%, H 8.16%, O 10.80%.
Description and references
Constituent of eucalyptus, myrrh, cassia, cumin, and other essential oils; prepared synthetically from p-isopropylbenzoyl chloride. Alternate synthesis by formylation of isopropylbenzene under pressure: Crounse, J. Am. Chem. Soc. 71, 1263 (1949). Toxicity study: P. M. Jenner et al., Food Cosmet. Toxicol. 2, 327 (1964).
Properties
Colorless to yellowish, oily liq; strong persistent odor; acrid, burning taste. d20 0.978. bp760 235-236°; bp35 131-135°. nD20 1.5301. Practically insol in water. Sol in alc, ether. LD50 orally in rats: 1390 mg/kg (Jenner).Derivative
Thiosemicarbazone.Nomenclature
CAS number: 3811-20-9
SHCH-58. C11H15N3S; mol wt 221.32.
C 59.70%, H 6.83%, N 18.99%, S 14.49%.
Description and references
Prepn: P. P. T. Sah, T. C. Daniels, Rec. Trav. Chim. 69, 1545 (1950); J. Bernstein et al., J. Am. Chem. Soc. 73, 906 (1951).