Nomenclature
CAS number: 135-20-6
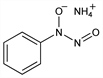
N-Hydroxy-
N-nitrosobenzenamine ammonium
salt;
N-nitrosophenylhydroxylamine ammonium salt.
C
6H
9N
3O
2; mol wt 155.15.
C 46.45%, H 5.85%, N 27.08%, O 20.62%.
Description and references
Prepd from phenylhydroxylamine by treating
with NaNO2 at 0° in presence of HCl, filtering, dissolving
in ether and treating with NH3, or by treating an ether
soln of phenylhydroxylamine with butyl or amyl nitrite and NH3 in the cold: Marvel, Kamm, Org.
Synth. 4, 19 (1925).
Properties
Crystals, mp 163-164°. Freely sol in water or alc. Keep in well-closed containers to which a piece of ammonium
carbonate has been added.Caution
This substance is reasonably anticipated
to be a human carcinogen: Report on Carcinogens,
Eleventh Edition (PB2005-104914, 2004) p III-73.Use
As a reagent for separating Sn from Zn, and Cu and
Fe from other metals. Ppts iron quantitatively from strongly acid
soln; as a quantitative reagent for vanadates with which it gives
a dark-red ppt sol in alkali soln, and for Ti with which it forms
a yellow ppt; also suitable for the colorimetric estimation of Al.
