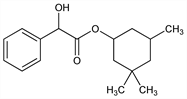
2704. Cyclandelate
Nomenclature
Description and references
Prepn: N. Brock et al., Arzneim.-Forsch. 2, 165 (1952); A. B. H. Funcke et al., ibid. 3, 503 (1953); van Sluis, Chem. Prod. 17, 375 (1954); GB 707227; W. T. Nauta, US 2707193 (1954, 1955 both to Brocades-Stheeman). Purification: D. Flitter, US 3663597 (1972 to Am. Home Prods.). Pharmacokinetics: A. Orr, J. R. Whittier, Int. J. Nucl. Med. Biol. 1, 205 (1974). GLC determn in human plasma: G. Andermann, M. Dietz, J. Chromatogr. 223, 365 (1981). Clinical trial in chronic cerebrovascular disease: S. Bassi et al., Br. J. Clin. Pract. 38, 344 (1984). Review of efficacy as cerebral and peripheral vasodilator: C. B. Blakemore, ibid. 34, Suppl., 3-9 (1984). Symposium on calcium modulation and clinical effects: Drugs 33, Suppl. 2, 1-141 (1987). Comprehensive description: C. M. Shearer, Anal. Profiles Drug Subs. Excip. 21, 149-168 (1992).
Properties
Crystals, mp 55.0-56.5°. bp14 192-194°. uv max (ethanol): 269, 258, 251 nm (ε 1575, 2020, 1630). Very sol in chloroform, methanol; freely sol in acetonitrile, ethyl acetate, DMF, toluene. Insol in water.Therapeutic Category
Vasodilator (peripheral, cerebral).
Keywords
Vasodilator (Cerebral); Vasodilator (Peripheral)