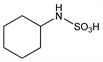
2703. Cyclamic Acid
Nomenclature
CAS number: 100-88-9
Cyclohexylsulfamic acid; cyclohexanesulfamic acid; Hexamic Acid (Abbott). C6H13NO3S; mol wt 179.24.
C 40.21%, H 7.31%, N 7.81%, O 26.78%, S 17.89%.
Description and references
Prepn: Audrieth, Sveda, US 2275125 (1942 to du Pont); J. Org. Chem. 9, 89 (1944); Thompson, US 2800501 (1957 to du Pont); Shah, Bernsen, US 3361799 (1968 to Abbott). Prepn of the Na salt: Robinson, US 2383617 (1945 to du Pont). Other prepns and metabolism: See Calcium Cyclamate. Sweetness of the sodium salt discovered by Michael Sveda at the University of Illinois in 1937. Toxicity: Taylor et al., Food Cosmet. Toxicol. 6, 313 (1968). Review of long-term toxicity and carcinogenicity of sodium cyclamate in mice: Brantom et al., Food Cosmet. Toxicol. 11, 735 (1973).
Properties
Sweet-sour crystals. mp 169-170°. Fairly strong acid. Very sparingly soluble in water. Slowly hydrolyzed by hot water.Derivative
Sodium salt.Nomenclature
Sodium cyclohexylsulfamate; cyclamate sodium; sodium cyclamate; Assugrin; Sucaryl Sodium; Sucrosa.C6H12NNaO3S; mol wt 201.22.
C 35.81%, H 6.01%, N 6.96%, Na 11.43%, O 23.85%, S 15.94%.