2711. Cyclobarbital
Nomenclature
CAS number: 52-31-3
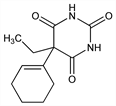
5-(1-Cyclohexen-1-yl)-5-ethyl-2,4,6(1H,3H,5H)-pyrimidinetrione; 5-(1-cyclohexen-1-yl)-5-ethylbarbituric acid; cyclobarbitone; hexemal; tetrahydrophenobarbital; Cavonyl; Cyclodorm; Cyklodorm; Fanodormo; Irifan; Namuron; Palinum; Phanodorm (Winthrop); Phanodorn (Winthrop); Philodorm; Pr"alumin; Pro-Sonil; Sonaform. C12H16N2O3; mol wt 236.27.
C 61.00%, H 6.83%, N 11.86%, O 20.31%.
Description and references
Prepn: GB 231150 (1924 to Bayer); Schulemann, Meisenburg, US 1690796 (1929 to Winthrop); Eckstein, Przem. Chem. 9, 390 (1953), C.A. 49, 11668c (1955). Toxicity data: G. Hofrichter et al., Arzneim.-Forsch. 17, 242 (1967).
Properties
Shiny crystals, insipid bitter taste, mp 171-174°. Very slightly sol in cold water, appreciably sol in boiling water. One gram dissolves in 5 ml alcohol, 20 ml ether. LD50 in mice, rats (mg/kg): 350, 290 i.p. (Hofrichter).Derivative
Calcium salt.Nomenclature
CAS number: 143-76-0
Hexemal calcium; Itridal; Kollerdormfix; Pronox (Reiss). C24H30CaN4O6; mol wt 510.60.
C 56.45%, H 5.92%, Ca 7.85%, N 10.97%, O 18.80%.
Properties
Minute crystals. Appreciably bitter taste. Slightly basic reaction. Soly in water about 1 part in 70 parts (w/w), in 95% alc about 1 part in 500 parts. Practically insol in ether, chloroform.Note
This is a controlled substance (depressant): 21 CFR, 1308.13.Therapeutic Category
Sedative, hypnotic.
Therapeutic Category (Veterinary)
Has been used as an anesthetic and sedative.Keywords
Sedative/Hypnotic; Barbituric Acid Derivatives