2715. Cyclobutyrol
Nomenclature
CAS number: 512-16-3
α-Ethyl-1-hydroxycyclohexaneacetic acid; 1-cyclohexanol-α-butyric acid; α-(1-hydroxycyclohexyl)butyric acid; 1-hydroxy-α-ethylcyclohexylacetic acid. C10H18O3; mol wt 186.25.
C 64.49%, H 9.74%, O 25.77%.
Description and references
Prepd by hydrolysis of the ethyl ester obtained by the Reformatski reaction: Kon, Naravanan, J. Chem. Soc. 1927, 1536; Kandiah, Linstead, ibid. 1929, 2150; J. G. Maillard et al., Bull. Soc. Chim. Fr. 1958, 244; eidem, DE 1094254; eidem, US 3065134 (1959, 1962 to Lab. Jacques Logeais). Activity studies: Roquet, Jousse, Arch. Int. Pharmacodyn. 125, 172 (1960).
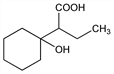
Properties
Colorless crystals from ether-petr ether, mp 81-82°. Formerly reported as viscous oil. d418.8 1.0010. bp24 164°; bp16 167-170°; nD18.8 1.4680 (Kon, Naravanan, loc. cit.). Slightly sol in water, petr ether. Very sol in alcohols, acetone, dioxane, chloroform, ether. Completely sol in aq alkalies at pH 7.5-8.Derivative
Sodium salt.Nomenclature
CAS number: 1130-23-0
Bilimix (Gero); Bis-Bil (Isola-Ibi); Colepan (Panther-Osfa); Dimene; Epa-Bon (Sierochimica); Hebucol; Maricolene; Tachicol (Radiumfarma); Tri-Bil (Biologici); Tribilina (Drumond). C10H17NaO3; mol wt 208.23.
C 57.68%, H 8.23%, Na 11.04%, O 23.05%.
Properties
White crystalline, slightly hygroscopic powder, mp 299-300°.Therapeutic Category
Choleretic.
Keywords
Choleretic