2718. Cyclodextrins
Nomenclature
Cycloamyloses; cycloglucans; Schardinger dextrins.Description and references
Naturally occurring, cyclic α-(1,4)-linked oligosaccharides of α-d-glucopyranose having a conical structure with a hollow, hydrophobic internal cavity and a hydrophilic outer surface. Form noncovalent inclusion complexes with a wide range of polar and apolar compounds. Originally obtained by the action of Bacillus macerans amylase on starch; α-, β- and γ-cyclodextrins are composed of six, seven and eight units, resp. Chemically modified derivatives have also been prepared with expanded inclusion capacities. Isoln of β-cyclodextrin: A. Villiers, Compt. Rend. 112, 536 (1891); of α- and β-cyclodextrin: F. Schardinger, Z. Untersuch. Nahr. Genussm. 6, 865 (1903); of γ-cyclodextrin: K. Freudenberg, R. Jacobi, Ann. 518, 102 (1935). Overview of chemistry: J. Szejtli, Chem. Rev. 98, 1743-1753 (1998); of production using cyclodextrin glycosyl transferase: A. Biwer et al., Appl. Microbiol. Biotechnol. 59, 609-617 (2002). Review of use in separation methods: E. Schneiderman, A. M. Stalcup, J. Chromatogr. B 745, 83-102 (2000); in cosmetics: H. Buschmann, E. Schollmeyer, J. Cosmet. Sci. 53, 185-191 (2002). Review of applications in drug delivery: R. Challa et al., AAPS PharmSciTech 6, E329-E357 (2005); T. Loftsson et al., Expert Opin. Drug Deliv. 2, 335-351 (2005).
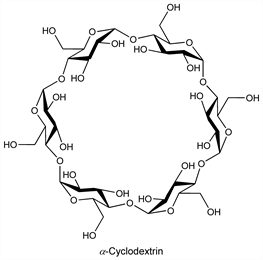
Derivative
α-Cyclodextrin.Nomenclature
Description and references
Total synthesis: Y. Takahashi, T. Ogawa, Carbohydr. Res. 164, 277 (1987).