10369. β-Cyclodextrin Sulfobutyl Ether Sodium Salt
Nomenclature
Description and references
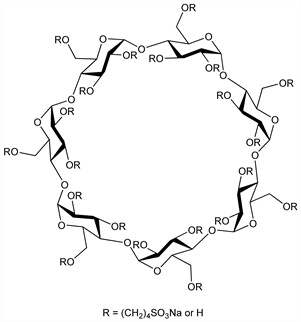
Polyanionic sulfobutyl ether deriv of β-cyclodextrin, q.v.; composite mixture of variably substituted isomers of the general formula C42H70-nO35.(C4H8SO3Na)n where n, the average degree of substitution, ranges between 6 and 7; mol wt ≈2163. Excipient that complexes with otherwise insoluble drugs and enhances their solubility and stability. Prepn and pharmaceutical applications: V. Stella, R. Rajewski, US 5134127; eidem, US 5376645 (1992, 1994 both to University of Kansas); and pharmaceutical formulations: V. Stella et al., US 5874418 (1999 to CyDex). Effect on drug solubility and stability: H. Ueda et al., Drug Dev. Ind. Pharm. 24, 863 (1998) DOI PubMed. Osmotic properties: E. A. Zannou et al., Pharm. Res. 18, 1226 (2001) DOI PubMed; V. J. Stella et al., J. Inclusion Phenom. Macrocyclic Chem. 44, 29 (2002) DOI. Characterization by capillary electrophoresis: R. J. Tait et al., J. Pharm. Biomed. Anal. 10, 615 (1992) DOI PubMed. Analysis by chromatography and mass spectrometry: S. Grard et al., J. Chromatogr. A 925, 79 (2001) DOI PubMed. Review of pharmacokinetics, safety, and toxicology: V. J. Stella, Q. He, Toxicol. Pathol. 36, 30-42 (2008) DOI PubMed.