Nomenclature
CAS number: 66-81-9
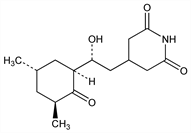
4-[(2
R)-2-[(1
S,3
S,5
S)-3,5-Dimethyl-2-oxocyclohexyl]-2-hydroxyethyl]-2,6-piperidinedione; 3-[2-(3,5-dimethyl-2-oxocyclohexyl)-2-hydroxyethyl]glutarimide; actidione; naramycin A; NSC-185; U-4527.
C
15H
23NO
4; mol wt 281.35.
C 64.03%, H 8.24%, N 4.98%, O 22.75%.
Description and references
Glutarimide antibiotic produced by Streptomyces
griseus; inhibits protein synthesis in eukaryotic cells. Isoln:
B. E. Leach et al., J. Am. Chem.
Soc. 69, 474 (1947); J. H. Ford, B. E. Leach, ibid. 70, 1223 (1948); A. J. Whiffen et al., US 2574519 (1951 to Upjohn). Production, assay and activity:
A. J. Whiffen, J. Bacteriol. 56, 283 (1948). Structure: E. C. Kornfeld et al., J. Am. Chem. Soc. 71, 150 (1949). Absolute configuration: F. Johnson et al., J. Am. Chem. Soc. 87, 4612 (1965). Total synthesis of dl- and l-forms: F. Johnson et al., ibid. 88, 149 (1966). Effect on protein synthesis: T. G.
Obrig et al., J. Biol. Chem. 246, 174 (1971). Use in selective recovery of Campylobacter
sp. from contaminated foods: M. P. Doyle, D. J. Roman, Appl. Environ. Microbiol. 43,
1343 (1982); in detection of Legionella sp. in water systems:
H. Inoue et al., Biocontrol Sci. 11, 69 (2006).
Properties
Crystals from water + methanol, mp 119.5-121° (Kornfield); also reported
as colorless plates from amyl acetate, mp 115-116.5° (Leach). Poisonous; skin and eye irritant. [α]D29 -3.38° (c = 9.47
in methanol); [α]D25 +6.8° (c = 2 in H2O). pKa 11.2.
Soly at 2°: water 2.1 g/100 ml, amyl acetate 7 g/100 ml. Also sol
in chloroform, ether, acetone, methanol, ethanol, other common organic
solvents except satd hydrocarbons. Relatively heat-stable, acid-stable,
destroyed by boiling in aq soln at pH 7 for 1 hr, but shows no loss
of activity after 15 min boiling. At pH 2 it is not destroyed by
boiling for 1 hr. Rapidly inactivated at room temp by dil alkali
with the formation of a volatile, fragrant ketone, 2,4-dimethylcyclohexanone.
Bitter taste, extremely repellent to rats. LD50 i.v. in mice: 150 mg/kg (Leach).Derivative
Acetate.
C
17H
25NO
5; mol wt 323.38.
C 63.14%, H 7.79%, N 4.33%, O 24.74%.
Properties
Glistening plates from methanol, mp 148-149°. [α]D25 +22° (c = 2.3 in
methanol).Use
Fungicide. In selective microbiological media.
As protein synthesis inhibitor in biomedical research.
