Nomenclature
CAS number: 108-91-8
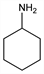
Cyclohexanamine; aminocyclohexane; hexahydroaniline.
C
6H
13N; mol wt 99.17.
C 72.67%, H 13.21%, N 14.12%.
Description and references
Prepd by the catalytic hydrogenation of aniline
at elevated temps and pressures. Fractionation of the crude reaction
product yields cyclohexylamine, unchanged aniline, and a high-boiling
residue contg N-phenylcyclohexylamine (cyclohexylaniline)
and dicyclohexylamine. Review and bibliography: Carswell, Morrill, Ind. Eng. Chem. 29, 1247 (1937).
Toxicity data: H. F. Smyth et al., Am. Ind. Hyg. Assoc. J. 30, 470 (1969).
Properties
Liquid. Strong, fishy, amine odor. d2525 0.8647.
mp -17.7°. bp760 134.5°; bp500 118.9°; bp300 102.5°; bp100 72.0°; bp50 56.0°; bp30 45.1°; bp25 41.3°; bp20 36.4°; bp15 30.5°. nD25 1.4565. Corrosive,
flammable. Strong base. Completely misc with water and
with common organic solvents, including alcohols, ethers, ketones,
esters, aliphatic hydrocarbons, aromatic hydrocarbons and their chlorinated
derivatives. On distillation with water, cyclohexylamine forms an
azeotropic mixture contg 44.2% cyclohexylamine by weight, bp760 96.4°. Reacts with excess ammonia and zinc chloride at 350° to produce α-picoline:
Nordt, Conversion of Hexahydroaniline into Pyridine Bases, Off. Pub. Bd., Report PB 704 (1941). LD50 orally in rats: 0.71 ml/kg (Smyth).Caution
Potential symptoms of overexposure
are irritation of eyes, skin, mucous membranes, respiratory system;
eye, skin burns; skin sensitization; cough, pulmonary edema; drowsiness,
lightheadedness, dizziness; diarrhea, nausea, vomiting. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 84.Use
In organic synthesis, manuf insecticides, plasticizers,
corrosion inhibitors, rubber chemicals, dyestuffs, emulsifying agents,
dry-cleaning soaps, acid gas absorbents.
