2751. Cycloserine
Nomenclature
Description and references
Antibiotic substance produced by Streptomyces garyphalus sive orchidaceus: Kuehl, Jr., et al., J. Am. Chem. Soc. 77, 2344 (1955); Hidy et al., ibid. 2345; Shull, Sardinas, Antibiot. Chemother. 5, 398 (1955); Shull et al., US 2773878 (1956 to Pfizer); Harned, US 2789983 (1957 to Commercial Solvents); GB 768007 (1957 to Commercial Solvents), C.A. 51, 10847e (1957); US 3124590 (1964 to Commercial Solvents); Howe, US 2845433 (1958 to Merck & Co.). Synthesis: Stammer et al., J. Am. Chem. Soc. 77, 2346 (1955); Peck, US 2772280 (1956 to Merck & Co.); Plattner et al., Helv. Chim. Acta 40, 1531 (1957); Holly, Stammer, US 2840565 (1958 to Merck & Co.). Prepn of crystalline calcium and magnesium salts: Harris et al., US 2832788 (1958 to Merck & Co.). HPLC determn in plasma and urine: D. G. Musson et al., J. Chromatogr. 414, 121 (1987). Comprehensive description: J. W. Lamb, Anal. Profiles Drug Subs. 1, 53-64 (1972); H. A. El-Obeid, A. A. Al-Badr, ibid. 18, 567-597 (1989).
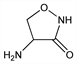
Properties
Crystals, decomp 155-156°. [α]D23 +116° (c = 1.17); [α]25546 +137° (c = 5 in 2N NaOH). uv max: 226 nm (E1%1cm 402). Sol in water, slightly sol in methanol, propylene glycol. Aq solns have a pH around 6. Forms salts with acids and bases. Neutral or acid solns are unstable. Aq solns buffered to pH 10 with sodium carbonate can be stored without loss for one week at refrigerator temps.Therapeutic Category
Antibacterial (tuberculostatic).
Keywords
Antibacterial (Antibiotics); Antibacterial (Tuberculostatic)