2752. Cyclosporins
Description and references
Group of nonpolar cyclic oligopeptides with immunosuppressant activity; produced by Tolypocladium inflatum Gams (formerly designated as Trichoderma polysporum [Link ex Pers.] Rifai) and other fungi imperfecti. Cyclosporins A through Z have been identified with A being the major component. Characterized by a C9-amino acid, MeBmt, q.v., cyclosporins bind to the cytosolic protein, cyclophilin, to form an active complex which inhibits the enzyme, calcineurin, a key component of T-cell activation. Isoln of A and C, structure of A: A. Rüegger et al., Helv. Chim. Acta 59, 1075 (1976); structure of C: R. Traber et al., ibid. 60, 1247 (1977). Production of A and C: E. H"arri et al., US 4117118 (1978 to Sandoz). Review of isoln, structure elucidation, synthesis and bioactivity: A. von Wartburg, R. Traber, Prog. Med. Chem. 25, 1-33 (1988); of biosynthesis and mode of action: A. Lawen, ibid. 33, 53-97 (1996). Review of role of calcineurin on bioactivity of cyclosporins: K. A. Jorgensen et al., Scand. J. Immunol. 57, 93-98 (2003). HPLC separation of components from fungal fermentation samples: J. Tuominen et al., Rapid Commun. Mass Spectrom. 12, 1085 (1998). HPLC-MS determn in blood: B. Vollenbroeker et al., Transplant. Proc. 37, 1741 (2005).
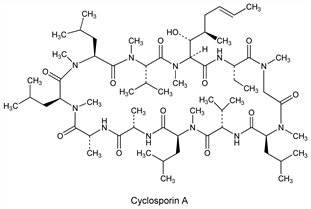
Derivative
Cyclosporin A.Nomenclature
Description and references
Comprehensive description: M. M. Hassan, M. A. Al-Yahya, Anal. Profiles Drug Subs. 16, 145-206 (1987). Toxicological evaluation: B. Ryffel et al., Arch. Toxicol. 53, 107 (1983). Symposium on therapeutic use in dermatology: J. Am. Acad. Dermatol. 23, part 2, 1241-1334 (1990). Review of use in canine skin diseases: E. Guaguere et al., Vet. Dermatol. 15, 61-74 (2004). Review of clinical experience in organ transplantation: C. J. Dunn et al., Drugs 61, 1957-2016 (2001); in treatment of dry eye: H. D. Perry, E. D. Donnenfeld, Expert Opin. Pharmacother. 5, 2099-2107 (2004).
Properties
White prismatic needles from acetone at -15°. mp 148-151°. [α]D20 -244° (c = 0.6 in chloroform); [α]D20 -189° (c = 0.5 in methanol). Sol in methanol, ethanol, acetone, ether, chloroform; slightly sol in water and saturated hydrocarbons. LD50 in mice, rats, rabbits (mg/kg): 107, 25, >10 i.v.; 2329, 1480, >1000 orally (Ryffel).Derivative
Cyclosporin B.Nomenclature
Properties
Amorphous white powder, mp 149-152°. [α]D20 -238° (c = 0.62 in chloroform). [α]D20 -168° (c = 0.56 in methanol).Derivative
Cyclosporin C.Nomenclature
Properties
Colorless prismatic needles from acetone at -15°. mp 152-155°. [α]D20 -255° (c = 0.5 in chloroform). [α]D20 -182° (c = 0.5 in methanol). Solubility similar to cyclosporin A.Derivative
Cyclosporin D.Nomenclature
Properties
Colorless prisms from acetone at -15°. mp 148-151°. [α]D20 -245° (c = 0.52 in CHCl3). [α]D20 -211° (c = 0.51 in methanol).Derivative
Cyclosporin G.Nomenclature
Description and references
Immunosuppressive profile: P. C. Hiestand et al., Transplant. Proc. 17, 1362 (1985); eidem, Immunology 55, 249 (1985).
Properties
Colorless, polyhedric crystals from ether + petroleum ether, mp 196-197°. [α]D20 -245° (c = 1.0 in chloroform). [α]D20 -191° (c = 1.04 in methanol).Caution
Cyclosporin A is listed as a known human carcinogen: Report on Carcinogens, Eleventh Edition (PB2005-104914, 2004) p III-75.Therapeutic Category
Cyclosporin A as immunosuppressant.
Therapeutic Category (Veterinary)
Cyclosporin A as immunosuppressant.Keywords
Cyclosporine: Immunosuppressant; Calcineurin Inhibitor