2769. Cyprenorphine
Nomenclature
CAS number: 4406-22-8
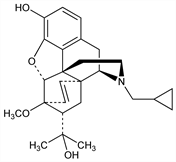
(5α,7α)-17-(Cyclopropylmethyl)-4,5-epoxy-3-hydroxy-6-methoxy-α,α-dimethyl-6,14-ethenomorphinan-7-methanol; N-(cyclopropylmethyl)-7,8-dihydro-7α-(1-hydroxy-1-methylethyl)-O6-methyl-6,14-endo-ethenonormorphine; N-(cyclopropylmethyl)-6,14-endo-etheno-7α-(2-hydroxy-2-propyl)tetrahydronororipavine; N-(cyclopropylmethyl)-6,14-endo-ethenotetrahydronororipavine. C26H33NO4; mol wt 423.54.
C 73.73%, H 7.85%, N 3.31%, O 15.11%.
Description and references
Narcotic antagonist closely related to diprenorphine, q.v. Prepn (7α- or 7β-linkage unspecified): BE 629070 (1963 to J. F. Macfarlan), C.A. 61, 13364g (1964); K. W. Bentley, US 3474101 (1969 to Reckitt & Sons); of 7α: K. W. Bentley, D. G. Hardy, J. Am. Chem. Soc. 89, 3281 (1967). Activity in rats: K. W. Bentley et al., Nature 206, 102 (1965). Use in large animals: M. R. Jainudeen, Vet. Rec. 89, 686 (1971); K. R. Presnell et al., J. Wildl. Dis. 9, 336 (1973). Binding to neuroleptic receptors: A. Czlonkowski et al., Life Sci. 22, 953 (1978). Determn by HPLC: I. Jane, A. McKinnon, J. Chromatogr. 323, 191 (1985).
Properties
Prisms from methanol, mp 234° (7α- or 7β-linkage unspecified).Derivative
Hydrochloride.Nomenclature
CAS number: 16550-22-4
M-285. C26H33NO4.HCl; mol wt 460.01.
C 67.89%, H 7.45%, N 3.04%, O 13.91%, Cl 7.71%.