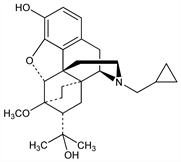
3340. Diprenorphine
Nomenclature
Description and references
Closely related to cyprenorphine, q.v. Prepn: K. W. Bentley, D. G. Hardy, J. Am. Chem. Soc. 89, 3281 (1967). Activity in rats: G. F. Blane, J. Pharm. Pharmacol. 19, 367 (1967); G. F. Blane, D. Dugdall, ibid. 20, 547 (1968). Use as etorphine antagonist in dogs: M. Grange et al., Rev. Med. Vet. 124, 899 (1973); in large animals: B. T. Alford et al., J. Am. Vet. Med. Assoc. 164, 702 (1974). Binding to opiate receptors: C. B. Pert et al., Life Sci. 16, 1849 (1975); J. Pearson et al., ibid. 26, 1047 (1980); to μ opiate receptors: J. J. Frost et al., ibid. 38, 1597 (1986). Treatment of experimental stroke in cats: D. S. Baskin et al., Neuropeptides 5, 307 (1985); eidem, J. Neurosurg. 64, 99 (1986). Determn by HPLC: I. Jane, A. McKinnon, J. Chromatogr. 323, 191 (1985). Toxicity data: N. S. Duggett et al., Toxicol. Appl. Pharmacol. 31, 141 (1977).