2786. Cytidine
Nomenclature
CAS number: 65-46-3
4-Amino-1-β-d-ribofuranosyl-2-(1H)-pyrimidinone; cytosine riboside; 1-β-d-ribofuranosylcytosine. C9H13N3O5; mol wt 243.22.
C 44.44%, H 5.39%, N 17.28%, O 32.89%.
Description and references
Constituent of nucleic acids. Isoln from yeast nucleic acid: Levene, Jacobs, Ber. 43, 3154 (1910); Levene, La Forge, ibid. 45, 608 (1912). Sepn from other nucleosides by ion-exchange chromatography: Cohn in Chargaff-Davidson, The Nucleic Acids vol. I (New York, 1955) p 211. Synthesis: Howard et al., J. Chem. Soc. 1947, 1052. Crystal structure: Furberg et al., Acta Crystallogr. 18, 313 (1965). Review: Basic Principles in Nucleic Acid Chemistry vol. 1, P. O. P. Ts'o, Ed. (Academic Press, New York, 1974) passim.
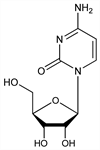
Properties
Long needles from 90% ethanol, dec 220-230°. [α]D25 +31° (c = 0.7 in water). Freely sol in water, less sol in alcohol. pK (amino, cationic) 4.22; pK (sugar, anionic) 12.5. uv max (pH 8.2): 271 nm (ε 9100); (pH 2.2): 280 nm (ε 13400), Voet et al., Biopolymers 1, 193 (1963).Derivative
Sulfate.(C9H13N3O5)2.H2SO4; mol wt 584.51.
C 36.99%, H 4.83%, N 14.38%, O 38.32%, S 5.49%.