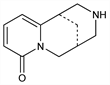
2789. Cytisine
Nomenclature
CAS number: 485-35-8
(1R)-1,2,3,4,5,6-Hexahydro-1,5-methano-8H-pyrido-[1,2-a][1,5]diazocin-8-one; baptitoxine; sophorine; ulexine; Cytiton (USSR). C11H14N2O; mol wt 190.24.
C 69.45%, H 7.42%, N 14.73%, O 8.41%.
Description and references
Toxic principle in seed of Laburnum anagyroides Medik. and other Leguminosae. Extraction: Ing, J. Chem. Soc. 1931, 2200; Sp"ath, Galinovsky, Ber. 65, 1526 (1932); 66, 1338 (1933); Lecoq, Bull. Soc. Chim. Fr. 10, 153 (1943). Structure: Ing, J. Chem. Soc. 1932, 2778. Synthesis: Bohlmann et al., Angew. Chem. 67, 708 (1955); Van Tamelen, Baran, J. Am. Chem. Soc. 77, 4944 (1955). Absolute configuration: Okuda et al., Chem. Ind. (London) 1961, 1751. Pharmacological properties: R. B. Barlow, L. J. McLeod, Br. J. Pharmacol. 35, 161 (1969).
Properties
Orthorhombic prisms from acetone, mp 152-153°. Sublimes. bp2 218°. [α]D17 -120°. pK1 6.11; pK2 13.08. Sol in 1.3 parts water, 13 parts acetone, 1.3 parts methanol, 3.5 parts alcohol, 30 parts benzene, 10 parts ethyl acetate, 2.0 parts chloroform. Practically insol in petr ether. LD50 in mice (mg/kg): 1.73 i.v.; 9.4 i.p.; 101 orally (Barlow, McLeod).Derivative
Hydrochloride.C11H14N2O.HCl; mol wt 226.70.
C 58.28%, H 6.67%, N 12.36%, O 7.06%, Cl 15.64%.
Properties
Deliquescent crystals, sol in water and alcohol. pH of 0.1 molar aq soln 4.3.Derivative
Nitrate monohydrate.C11H14N2O.HNO3.H2O; mol wt 271.27.
C 48.70%, H 6.32%, N 15.49%, O 29.49%.