2822. Dapsone
Nomenclature
Description and references
Prepn: FR 829926 (1938 to I. G. Farbenind.), C.A. 33, 1761 (1939); Buckles, J. Chem. Educ. 31, 36 (1954); Ferry et al., Org. Synth. coll. vol. III, 239 (1955). Antibacterial action and metabolism: J. Francis, A. Spinks, Br. J. Pharmacol. 5, 565 (1950). Mechanism of toxic action: Wu, DuBois, Arch. Int. Pharmacodyn. Ther. 183, 36 (1970). Comprehensive description: C. E. Orzech et al., Anal. Profiles Drug Subs. 5, 87-114 (1976). Review of pharmacology and therapeutic use: J. Uetrecht, Clin. Dermatol. 7, 111-120 (1989). Clinical trial with chlorproguanil, q.v., in drug-resistant malaria: T. Mutabingwa et al., Lancet 358, 1218 (2001).
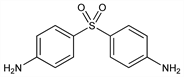
Properties
Crystals from 95% ethanol. mp 175-176° (also a higher melting form, mp 180.5°). pKb 13.0. Soluble in alcohol, methanol, acetone, dil hydrochloric acid. Practically insol in water.Use
Hardening agent in the curing of epoxy resins.Therapeutic Category
Antibacterial (leprostatic); in treatment of dermatitis herpetiformis.
Therapeutic Category (Veterinary)
Antibacterial; coccidostat.Keywords
Antibacterial (Synthetic); Sulfones; Antibacterial (Leprostatic); Dermatitis Herpetiformis Suppressant